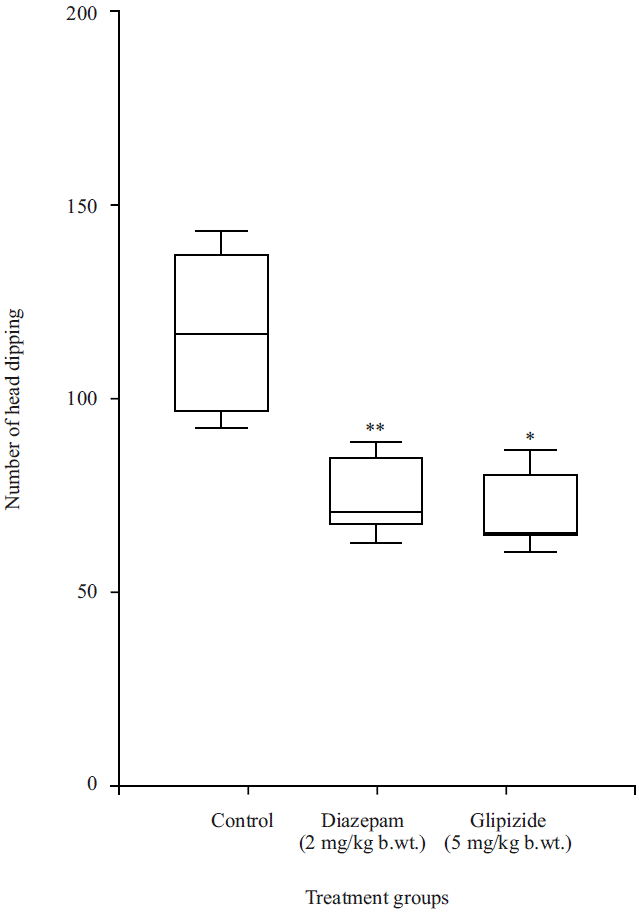ABSTRACT
Background and Objective: Nowadays, sulfonylurea including Glipizide have been tested to manage diabetes. However, their safety index yet not extensively studied. Our research aims were to assess neuro and the major organs' safety of Glipizide while their effects on different lipid and other biomedical parameters have been also tested. Materials and Methods: Streptozotocin (STZ)-induced T2DM in male swiss Albino mice model has been developed on twenty rats and assigned to four different groups to receive their treatments. At the end of 7 days treatment protocol, the neurological study was carried out by open field, hole board, forced swimming, dark and lighthouse and elevated plus-maze test. After sacrifice, major organs weight and serum level of high-density lipoprotein cholesterol (HDL-C), low-density lipoprotein cholesterol (LDL-C), very-low-density lipoprotein cholesterol (VLDL-C), total cholesterol (TC) and triglyceride (TG) have been evaluated. Results: Glipizide produces some neurological effects in almost all laboratories set up except elevated plus-maze test, whereas an effect on the whole board test was found better than standard diazepam. Present data replicate the significant downflow for plasma glucose and body weight gain over the treatment period while also having minimal effect on major organ weight and fat deposition. It also exposed that Glipizide also has some suppressing effects on TC, TG, LDL-C, VLDL-C levels compared to the control group. Conclusion: Present findings bring a clear projection that Glipizide at a dose of 5 mg kg–1 generates some anxiolytic activity with a remarkable plasma, glucose level and bodyweight reduction and minimal effects on lipid profile and major organs weight variation.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/jms.2023.7.17
URL: https://scialert.net/abstract/?doi=jms.2023.7.17
INTRODUCTION
Diabetes mellitus (T2DM) is an insulin released abnormality, inappropriate effects of insulin, or even both, a metabolic condition that is identified by hyperglycemia1. This chronic hyperglycemic condition is described as huge damage, malfunction and degradation of different organs, such as the brain, heart and blood vessels1. There are two types of diabetes mellitus. Type 1 DM, known as autoimmune diabetes, occurred by the depletion of pancreatic β-cells2 and type 2 DM is known as a relative lacking insulin caused by the abnormalities of pancreatic β-cells and resistance of insulin in the target organs3. In 2019, the worldwide prevalence for diabetes is forecast at 9.3% (463 M), 10.2% (578 M) in 2030 and 10.9% (700 M) in 20454. The WHO reports that the 7th largest cause of death in 2016 was diabetes that was 1.6 M. In Bangladesh, the prevalence of diabetes in adults increased dramatically, from 4% in 1995 to 2000 and 5% between 2001 and 2005 to 9% between 2006 and 20105. More than 350 M people around the world will be estimated to have diabetes by 2030 according to estimates prepared by the World Health Organization6. The IDF (International Diabetes Federation) reports a prevalence of 13% in 20307.
Due to differences in glucose levels in diabetes patients, there is an interrelation between diabetes and anxiety/depression8. Lustman et al.9 showed that anxiety is linked to the impaired balance of glucose and Kan et al.10 showed that insulin resistance is related to depression. Grigsby et al.11. reported that 40% of patients suffering from diabetes have elevated anxiety symptoms and 14% of patients have a general incidence of anxiety disorders. Agrawal et al.12 showed that 45% of the psychological conditions in diabetes patients include depression and anxiety. The occurrence rates of depression in patients with type 1 diabetes could be up to three times higher, relative to the general population around the world and double that in people with type 2 diabetes13.
Lipid abnormalities usually occur in diabetes patients often called "Diabetic Dyslipidemia” characterized by the excessive level of TC, TG, LDL-C and VLDL-C particulates with alleviating HDL-C14. In people with T2DM and prediabetes, lipid abnormalities are usual15. Dyslipidemia in diabetes patients is more prominent because insulin resistance or dysfunction influences main lipid-metabolism enzymes and routes16. An excessive free discharge of fatty acid in insulin-resistant fat cells is one pathway that underlies this relation. Increased amounts of fatty-free acids help the development of triglycerides that accelerate apolipoprotein B (ApoB) and very-low-density lipoprotein (VLDL) cholesterol release as well. The elevated levels of ApoB and VLDL raise the risk of cardiovascular disorder (CVD)17. Besides high levels of ApoB and VLDL, high-density lipoprotein (HDL) cholesterol levels are also correlated with hyperinsulinemia, an essential element of metabolic syndrome that may be a distinct cause of cardiovascular disease18. The nature of lipid particles of any diabetic dyslipidemia patient has been suggested to become more atherogenic19 and there is a well-founded strong causative relationship between atherosclerosis as well as dyslipidemia20. About 70% population in the world aged over 65 with DM would die from cardiovascular disease or stroke21. In T2DM, there is a link between cardiovascular disease and lipid profile22. Patients with T2DM have an increased probability of coronary artery disease, the leading risk factor in T2DM patients, which is double to four folds higher23. In diabetes, better glycemic management provides positive feedback on lipoprotein levels, decreasing the level of cholesterol and triglyceride by lowering the distribution of very low lipoprotein density (VLDL) and increasing LDL disruptions by minimizing glycation and LDL receptor uprisings24.
To control glucose levels in type 2 DM patients, Glipizide, a second-generation sulfonylurea drug, was formulated in Italy in 1970 and approved for medical use in the United States by the food and drug administration (FDA) in 198425. Among the other second-generation sulfonylurea, Glipizide shows a more potent hypoglycemic effect26. By partially binding with potassium channels, Glipizide sensitizing pancreatic beta cells induces beta-cell depolarization. This depolarization activates the voltage-gated calcium channels and facilitates the release of insulin from pancreatic beta cells26. In peripheral sites, it increases insulin sensitivity as well27.
There is no such data available to determine the Glipizide effect on anxiety/depression as well as physiological impact with lipid profile modification. Hence, our current research is designed to investigate the comparative effect of Glipizide and standard atorvastatin on hypoglycemic effect, bodyweight variation, lipid profile and the neuroprotective profile of both treatments in the model of diabetic swiss Albino mice.
MATERIALS AND METHODS
Study area: The study was conducted over July-December, 2020.
Experimental animals: Four to five weeks old male swiss Albino mice weighing approximately 25±5 g have been used and taken from the livestock house of the University of Jahangir Nagar (Savar), Dhaka, Bangladesh for this study. Feeding the mice was performed at 25±20°C and 55±10% relative humidity by ICDDRB formulated rodent food diet and water at Libitum. The animals were housed in clear polypropylene cages. For seven days before experimenting, they were permitted to familiarize themselves with laboratory conditions. The procedure employed in this research in the mice model was designed in compliance with the recommendations of the Committee on Institutional Animal Ethics28.
Collection of drugs: Active pharmaceutical ingredient (API) of Glipizide was acquired from BEXIMCO Pharmaceuticals Ltd, Bangladesh. API of standard drugs Atorvastatin (40 mg) and diazepam (2 mg) were kindly given by SQUARE Pharmaceuticals Ltd., Bangladesh. In this study, the analytical grade was maintained for all other reagents and materials.
Study design: Precisely 20 healthy mice were arbitrarily chosen for the study and divided into four groups of 5 mice each once streptozotocin (STZ)-induced T2DM established in mice. In detail, all mice were fasted overnight for at least 12 hrs and the use of STZ (4 weeks followed by an intraperitoneal injection of STZ (35 mg kg–1 suspended in 0.1 mol L–1 citrate buffer at pH 4.5) mediated hyperglycemia in each fasted mice29,30. Meanwhile, the early phase of hypoglycemia conditions was maintained by oral administration of 10% dextrose. Blood glucose was measured by the glucose oxidase technique with a Glucometer after 48 hrs of STZ administration. The following treatment only comprised those mice which were developed hyperglycemia (glucose>250 mg dL–1):
| • | Group 1 (Control group): All diabetic mice fed standard laboratory diet and water in Libitum |
| • | Group 2 (Atorvastatin group): All diabetic mice fed atorvastatin with the daily diet as a requirement of 40 mg kg–1 b.wt. |
| • | Group 3 (Diazepam group): All diabetic mice fed diazepam with the daily diet as a requirement of 2 mg kg–1 b.wt. |
| • | Group 4 (Glipizide group): All diabetic mice fed Glipizide with the daily diet as a requirement of 5 mg kg–1 b.wt. |
At the end of the 7 days treatment, mice of groups 1, 3 and 4 were evaluated in different neurological treatment set up to examine the treatment effects on the neurological system.
Neurological evaluation
Open field test: The open-field test was carried out as a reference to a method defined by Farooq and Michael31 for an evaluation of locomotor activity, exploration and anxiolytic attitude. This was achieved in 30 minutes by counting the number of squares crossed with all four paws. Control behaviours and drug-treated mice were controlled to prevent the impact of order in a balanced way.
Hole board: A Head dip box is used to check the anxiolytic behaviour of mice. In this experiment, a specially built square box formed with many holes at the bottom was used to count the deep number of the heads of the animal at a given time. Both control and drug-treated animals were put independently in the head dip box. This head dipping behavioural experiment was conducted using File and Wardill reference methods32.
Forced swim test: To determine the antidepressant function of test animals, the forced swim test is one of the most popular experimental models. This test was performed according to the previously developed method of Porsolt33. Mice were put in the specially built plastic cylinder with water at room temperature up to a marked amount individually for 6 min. Mice immediately shift their front and hind paws when put in water. With the aid of a stopwatch, the activity period of the animal is determined by 4 min in 6 min.
Elevated plus maze test: Elevated plus maze test is commonly recognized for the assessment of the anxiolytic effects of pharmacologically active components. This test was performed in conjunction with the Lister reference system34. Each mouse was put in the centre of the maze in front of a closed arm. Four paws in one arm were identified as entries in the arm. The following parameters were then noticed and counted: (a) Numbers of entries, (b) Time spent in closed and open arms for 5 min. The percentage of time that each animal spent on the open arm was measured as follows:
Dark-light house test: The dark-light house test is one of the instruments designed for Neuropharmacological behaviour testing in mice. This apparatus is composed of a two-compartment plastic case, one two-thirds area is illuminated and the other one-third area is dark. The number of entries and time consumed in each compartment is reported for 5 min for each animal positioned in the centre of the transparent or illuminated area. This test was conducted using Jacqueline and Frederick's reference system35.
Evaluation of lipid profile
Animal sacrifice and pathological examination of organ weight: After finishing 7 days of treatment, the experimental animals were thoroughly anaesthetized before sacrificing with the proper dose of chloroform. The blood samples were obtained in the centrifuge tube by cardiac puncture after sacrifice. The liver and kidney and the accumulated fat were detached from the carcasses after taking samples of blood in the Eppendorf and then stored in normal saline. After the organ was collected, every test animal’s organ was weighed individually and its average group weight was evaluated statistically.
Determination of serum cholesterol, triglyceride, HDL, LDL, VLDL level: Serum was collected from previously obtained blood samples of experimental mice through the centrifugation process and preserved at -80°C until further analysis. Then the enzyme method utilized Auto Pack kits were used to measure TC (total cholesterol), TG, HDL, LDL and VLDL. The traditional company Human Diagnostics Worldwide, Germany developed these reagents. The enzymatic colourimetric (endpoint) method was used for the determination of total serum cholesterol (TC)36 while serum triglyceride (TG) was measured with enzymatic colourimetric GPO-PAP method37 and HDL (High-Density Lipoprotein) which was assessed with Mindray BA-88A Semi-Auto Cline Chemistry Analyzer (Crown Healthcare, Tanzania) through phosphotungstate method38. Triglyceride values (VLDL = Triglyceride/5) were used to obtain VLDL (Very Low-Density Lipoprotein) value. Based on the equation of the Friedewald, LDL cholesterol was achieved39:
LDL cholesterol (mg dL–1) = Total cholesterol-(HDL+VLDL) |
Statistical analysis: To carry out a statistical evaluation of the findings One-way Analysis of Variance (ANOVA) guided by Dunnett’s post hoc test was used as necessary. The data were expressed as Mean±SEM (n = 5). All statistical data were analyzed using by statistical package for social science (SPSS) version 20 software. There were considered significant variations among groups at the level of p<0.05.
RESULTS
Comparative neuroprotective status of glipizide and diazepam
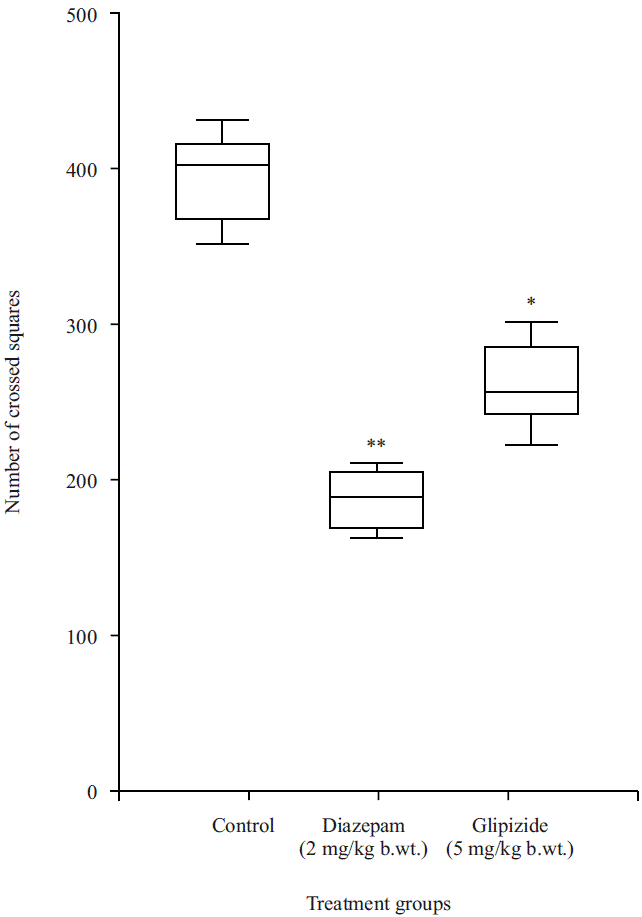
Open field test: Figure 1 illustrates the comparative anxiolytic behaviour of Glipizide and standard drug diazepam. The findings showed that Glipizide at 5 mg kg–1 dose significantly produced higher anxiolytic activity (263.0±13.09) by decreasing the number of squares crossed with all four paws in the open field when compared with the standard drug. It also expressed that the anxiolytic effect of Glipizide was more than the control group (395.8±13.82).
Hole board test: In the hole board test, the number of head dipping was reduced in the Glipizide treatment group 5 mg kg–1 after 30 min oral administration (65.0±18.17) and the effect was more than the standard diazepam as well as much more compared to the control group (117.40±9.97).
| Fig. 1: | Anxiolytic effect of Glipizide and diazepam in the open field test All values are expressed as Mean±SEM (n = 5), SEM = Standard error mean and significant level stated as *p<0.05 compared to control |
| Table 1: | Effect of Glipizide and diazepam on the forced swim test |
| Groups | Immobility time (sec) | Mobility time (sec) |
| Control (saline) | 110.80±3.48 | 129.20±2.41 |
| Diazepam (2 mg kg–1 b.wt.) | 62.80±3.74** | 177.0±3.64** |
| Glipizide (5 mg kg–1 b.wt.) | 82.0±3.78* | 158.0±3.72* |
| Values are expressed as Mean±SEM (n = 5), SEM = Standard error mean, significant level stated as *p<0.05 and **p<0.01 compared to control | ||
| Table 2: | Effect of Glipizide and diazepam in the elevated plus-maze test |
| Groups | Open arm time (sec) | % Open arm time (sec) | Open arm entries |
| Control (saline) | 39.75±5.02 | 13.25±1.67 | 7.60±1.40 |
| Diazepam (2 mg kg–1 b.wt.) | 65.11±20.54* | 21.70±6.85* | 8.80±1.66 |
| Glipizide (5 mg kg–1 b.wt.) | 43.01±2.83 | 15.67±0.94 | 7.78±1.12 |
| Values are expressed as Mean±SEM (n = 5), SEM = Standard error mean and significant level stated as *p<0.05 compared to control | |||
| Table 3: | Effect of Glipizide and diazepam in the dark and light house test |
| Groups | Time spent in light compartment (sec) | Number of entries in light compartment |
| Control (saline) | 77.63±16.01 | 9.60±1.12 |
| Diazepam (2 mg kg–1 b.wt.) | 103.26±26.77** | 11.20±1.56** |
| Glipizide (5 mg kg–1 b.wt.) | 91.96±8.89 | 12.60±1.54 |
| Values are expressed as Mean±SEM (n = 5), SEM = Standard error mean and significant level stated as **p<0.01 compared to control | ||
| Fig. 2: | Effect of Glipizide and diazepam in the hole board test All values are expressed as Mean±SEM (n = 5), SEM = Standard error mean, significant level stated as *p<0.05 and **p<0.01 compared to control |
However, the data were statistically not significant in Fig. 2.
Forced swim test: In the forced swim test, the following data expressed that Glipizide significantly (p<0.05) reduced the immobility time while also increasing the mobility time more than the control while less than the standard group when administered at a dose of 5 mg kg–1 b. wt., in Table 1. These findings showed a shred of evidence that the antidepressant effect of Glipizide is comparable to the standard diazepam.
Elevated plus-maze test: In the elevated plus-maze, Glipizide, administered at a dose of 5 mg kg–1 b.wt., decreased the time spent in the open arm (23.01±2.83) as well as the percentage of time spent in the open arm (7.67±0.94) when compared to control and diazepam group. It also observed that the number of entries in the open arm was less than both saline and diazepam treated groups in Table 2. From the outcome, it can easily determine that Glipizide imparts less anxiolytic activity than standard diazepam.
Dark and lighthouse test: Results obtained from dark and lighthouse test expressed that Glipizide 5 mg kg–1 not only increased the time spent in the light compartment (91.96±8.89) more than control (77.63±16.01) group but also elevated the number of entries in the light compartment (12.60±1.54) when compared with other groups in Table 3. These findings reveal that Glipizide has produced a few anxiolytic effects compared to diazepam treatment.
Evaluation of serum blood glucose: Table 4 shows that Glipizide significantly reduced serum blood glucose level (211.58±14.05) at the very first day of the treatment and the day of sacrifice whereas, at Cmax of Glipizide, the repression of blood glucose level was more noticeable (161.69±12.11) while compared to their initial, end day effect and data from the control group.
| Table 4: | Effect of atorvastatin and Glipizide on serum blood glucose level |
| Initial day reading | After 7 days of treatment reading | ||
| Groups | After 12 hrs fasting (mg dL–1) | After 12 hrs fasting (mg dL–1) | At Cmax of glipizide and atorvastatin (mg dL–1) |
| Control (saline) | 421.56±67.73 | 453.34±23.08 | |
| Atorvastatin (40 mg kg–1 b.wt.) | 438.85±58.84 | 495.94±21.40 | 503.31±22.62*** |
| Glipizide (5 mg kg–1 b.wt.) | 296.64±41.34 | 211.58±14.05*** | 161.69±12.11* |
| Values are expressed as Mean±SEM (n = 5), SEM = Standard error mean, significance level stated as ***p<0.001, **p<0.01 and *p<0.05 | |||
| Table 5: | Effect of atorvastatin and Glipizide on body weight |
| Groups | Initial day weight, mg | After 7 days of weight, mg | % of body weight gain or loss |
| Control (saline) | 28.03±0.63 | 32.94±0.35 | ↑17.37 |
| Atorvastatin (40 mg kg–1 b.wt.) | 27.41±2.29 | 22.60±0.50*** | ↓17.55*** |
| Glipizide (5 mg kg–1 b.wt.) | 26.53±0.5 | 22.86±2.48** | ↓13.83** |
| Values are expressed as Mean±SEM (n = 5), SEM = Standard error mean, significance level stated as ***p<0.001, **p<0.01 and *p<0.05 | |||
| Table 6: | Organ weight variation among mice of different experimental groups |
| Group | The average weight of the liver (g) | The average weight of the deposited fat (g) | The average weight of the right kidney (g) | The average weight of the left kidney (g) |
| Control | 2.048±0.128 | 0.373±0.025 | 0.287±0.022 | 0.264±0.020 |
| Glipizide (5 mg kg–1 b.wt.) | 2.473±0.079 | 0.358±0.176 | 0.306±0.042 | 0.299±0.038 |
| Atorvastatin (40 mg kg–1 b.wt.) | 2.106±0.182 | 0.306±0.146 | 0.322±0.077 | 0.298±0.068 |
| Values are expressed as Mean±SEM (n = 5) and SEM = Standard error mean | ||||
| Table 7: | Analysis of lipid profile of different experimental groups |
| Lipid profile parameter (mg dL–1) | Control group | Atorvastatin group (40 mg kg–1 b.wt.) | Glipizide group (5 mg kg–1 b.wt.) |
| TC | 71.03±5.17 | 64.29±6.46 | 69.23±6.77 |
| TG | 107.11±5.88 | 72.40±12.80 | 93.51±28.52 |
| HDL | 0.53±0.07 | 1.22±2.03 | 0.68±0.05 |
| LDL | 33.95±6.23 | 27.14±7.56 | 29.94±8.19 |
| VLDL | 21.42±1.18 | 14.48±2.53 | 18.71±2.98 |
| Values are expressed as the Mean±SEM (n = 5), SEM = Standard error mean, TC = Total Cholesterol, TG = Triglyceride, HDL = High-density lipoprotein, VLDL = Very low-density lipoprotein and LDL = Low density lipoprotein | |||
However, it was also seen that atorvastatin boosted serum blood glucose level and increased dramatically when the drug reached peak plasma concentration.
Analysis of histological parameters
Analysis of body weight: Bodyweight variation of different experimental groups was expressed in Table 5. Results showed that the bodyweight losing tendency was more significant in Glipizide (13.83%) and atorvastatin (17.55%) treatment groups when compared with the control group (17.37%). But the data determined that Glipizide has a more potent tendency to lose body weight than the standard atorvastatin group.
Analysis of organ weight variation: Results obtained from Table 6 showed that in comparison with the standard atorvastatin group, the average weight of the liver (2.473±0.079) was slightly increased in the Glipizide treatment group. Glipizide treatment also increased the average weight of both right (0.306±0.042) and left (0.299±0.038 g) kidneys more than the control group (0.287±0.022 and 0.264±0.020 g) when compared to the standard atorvastatin group (0.322±0.077 and 0.298±0.068 g). In the Glipizide treatment group, it was seen that low-fat (0.358±0.176 g) accumulation occurred in comparison with the control group (0.373±0.025 g). Findings indicated that no significant values were observed.
Biochemical examination: From Table 7 it can be expressed that Glipizide reduced serum total cholesterol (TC) (69.23±6.77 mg dL–1) and low-density lipoprotein (LDL) (29.94±8.19 mg dL–1) levels when compared with the control group (71.03±5.17 and 33.95±6.23 mg dL–1 respectively). Triglyceride (TG) (93.51±28.52 mg dL–1) and very-low-density lipoprotein (VLDL) (18.71±2.98 mg dL–1) levels were also decreased more by Glipizide treatment compared with the control group (107.11±5.88 and 21.42±1.18 mg dL–1 respectively). As expected, the level of high-density lipoprotein (HDL) was increased (0.68±0.05 mg dL–1) in the Glipizide treatment group more than in the control group (0.53±0.07 mg dL–1). All the findings for Glipizide treated group is comparable to the atorvastatin treated group.
DISCUSSION
In recent decades, diabetes mellitus and its related consequences, including diabetic neuropathy and dyslipidemia, have become a major phenomenon. Numerous research demonstrates that type 2 diabetes is strongly associated with hyperglycemia, dyslipidemia and cardiovascular dysfunction and contributes significantly to worldwide morbidity and death40. It is therefore important to seek effective antidiabetic drugs with fewer detrimental effects and an improved expectation of controlling diabetes. In our research, we examined the two most widely utilized antidiabetic drugs called Glipizide with comparative neurological as well as hyperlipidemic effects.
The primary type of neurological disorders is anxiety and depression. Depression is a severe but widespread condition, linked to high relapse and death rates41. A comorbidities level between diabetes and depression is predicted with diabetes and mental illness that predominantly influences 8.3 and 10% of the worldwide population, respectively42. The link between diabetes and mental illness has long been known43. Popescu et al.44 proposed that multiple conditions exist explicitly or implicitly in patients with T2DM on the central and peripheral nervous system. Roy et al.45. stated that depression in Bangladesh is widespread in ambulatory patients suffering from type 2 diabetes. Another research performed by de Groot et al.46 showed that patients with diabetes tend to suffer depression more likely than their mates without diabetes. But the neurobiological basis for this disease production remains obscure47. The research demonstrated that this could be linked to the presence of noradrenergic, serotonergic, glutaminergic and GABAergic neuron and hormone transmitters and transporters48. A low level of GABA, an inhibitory neurotransmitter, in the central nervous system (CNS) leads to anxiety dysfunction with impaired functions or decreased expression of its main Gamma-Aminobutyric Acid (GABAA) receptor49.
In this research, multiple test models were used to analyze Glipizide and standard drug diazepam effects on the neurological system. In the open field test, the locomotor activity measures the excitatory level of the CNS50 and any decline in this function can be closely correlated with the sedation due to depression of the CNS51. In this test Glipizide significantly reduced the locomotor's activity than the control group (Fig. 1). The more head dips in the hole the more anxiolytic behaviour is involved in the hole board test52. This test easily tests the animal's reaction to a strange surrounding and enables the observation of various behaviour52. Here, the number of head dipping was reduced more by Glipizide treatment than the control group (Fig. 2). The dark lighthouse test suggests that a longer time spent in the light compartment explains the anxiolytic behaviour of the given substance53 and the EPM also describes the anxiolytic activity of any given sample by increasing the number of entries as well as time spent in the open arm of the EPM54. Results obtained from both the dark lighthouse test and the EPM test suggested that Glipizide produced a comparable anxiolytic effect with the diazepam group (Table 2 and 3). In animal models, a forced swim test is preferable to determine anti-depressant like behaviour. Anti-depressant behaviour is assessed by reducing the duration of immobility55. In this test, Glipizide produced more anti-depressant activity than control animals (Table 1). From the above test result, it can understand that Glipizide generates more anxiolytic and anti-depressant activity than control. The anxiolytic effect of Glipizide may be due to the more CNS hyperpolarization by binding with Gamma-Aminobutyric Acid (GABAA) receptor or benzodiazepine receptor triggered chloride channel. In the sulfonylurea class, identical effects were found in other generations56. No such study was carried out before to determine the exact mechanism of anxiolytic activity of Glipizide.
Blood glucose level disturbances are easily related to diabetes. Hyperglycemia is a frequent effect of T2DM57. In a group of individuals with type 2 diabetes, not only cognitive performance was compromised but also their mood condition was worsened during acute hyperglycemia58. The rising body weight is linked to insulin resistance that boots and the threat of cardiovascular disorder59. In the current study, it showed that Glipizide, 2nd generation sulfonylurea drug, significantly reduced serum blood glucose level even after reaching peak plasma concentration than the control group. A previous study also demonstrated that Glipizide treatment prominently reduced plasma blood glucose levels60. Arnouts et al.26 suggested that a much more non-polar side chain is available in the second-generation sulfonylurea, which results in hypoglycemic effects with greater power. Melander et al.61. proposed that Glipizide has the quickest absorption and action in contrast to other sulfonylurea’s. A previous study by Qaseem et al.62 showed that in obese patients of Type 2 DM, metformin produced better weight-losing activity instead of gaining than Glipizide. Another study from Kahn et al.63 demonstrated that the weight loss of metformin-treated diabetic patients was 2.7 kg for 4 years with rosiglitazone and glyburide, respectively showing weight elevation of 4.8 and 1.6 kg. Glipizide may induce cell depolarization when blocking the ATP dependent potassium channel that stimulates voltage-dependent calcium channel, which triggers the secretion of insulin and this insulin sensitizes peripheral target cells such as muscle, liver, kidney to take outside glucose and makes them fat like glycogen for storage26. Wang et al.64 demonstrated that metformin therapy may boost the cardiometabolic risk factor by decreasing body weight and body fat mass in patients with identified type 2 diabetes.
Dyslipidemia is a condition when the number of lipids increases in the blood that causes severe conditions such as atherosclerosis, which leads to coronary artery disease, hypertension and stroke. This hyperlipidemia is common in type 2 diabetes. The liver develops very-low-density lipoprotein (VLDL) extensively in a diabetic condition and it performs a vital role in increasing the number of triglycerides65. By altering the intra-hepatic destruction of Apo B-100, the densities of free fatty acid help to develop triglycerides in the liver through VLDL secretion66. Diabetic patients suffer from low plasma discharge of chylomicrons after ingestion of dietary fat. As a result, chylomicrons concentration is increased and the accumulation of fatty acid is also enhanced in the liver. The key reason for defending Apo B-100 against degradation, leading, in turn, to boost in synthesis and secretion of VLDL, is this deposited fatty acid. The reduction of insulin or insulin activity hampers the post-translational deterioration of Apo B-100 in the liver which facilitates more release of VLDL67.
Lecithin Cholesterol Acyltransferase (LCAT) is an enzyme that plays a significant role in converting free cholesterol into cholesteryl ester. This enzyme also helps to transform premature HDL to mature HDL. This mature HDL carries cholesteryl ester from blood and attaches to the liver by the SR-B1 receptor and then returns cholesteryl to the liver for excretion. Without LCAT, mature HDL isn’t found68. Research showed that a decline in coronary artery disease (CAD) is linked to increasing HDL-C levels69.
Diabetes induced hyperglycemia occurs due to an excessive increase of fat coming from adipose tissue through the utilization of glucose70. In diabetic conditions, hormone-responsive lipase enzyme typically disintegrates the accumulated triacylglycerol and turns into fatty acids. This fatty acid then transforms into phospholipids and cholesterols in the liver. The conjugation of phospholipids and cholesterol occurred with a surplus amount of TG produced in the liver can be released into the bloodstream in the form of lipoproteins. Also, lipoprotein lipase, known as the breakdown enzyme for triglycerides, activity is inhibited in diabetic conditions. As a result, elevated triglyceride levels are therefore noticed71.
Again, in a diabetic situation, insulin doesn't work properly on b-hydroxy-b-methylglutaryl Coenzyme-A (HMG-CoA) reductase, an enzyme potentially used for the breakdown of cholesterol72. Therefore, the reduction of intracellular cholesterol occurs and this reduction promotes the increase of hepatic LDL receptors which reduces the circulating LDL through binding with LDL receptor73.
In our study, we observed that Glipizide reduced the level of TC, TG, LDL-C and VLDL-C when compared to the control group where TG and VLDL-C levels are known to be possible bioindicators of cardiovascular disease progression. Triglycerides are independent risk factors for coronary heart diseases74. However, the reduction of TG levels may be due to an increase in endothelium bound lipoprotein lipase which regulates the disposal of lipids fuels in the body17. Cefalu et al.60 showed the effect of Glipizide on reducing TC and LDL-C levels in type 2 diabetes patients. Abba and Abbas75, showed that 2nd generation drugs of sulfonylurea’s remarkably reduced the level of TC, TG and LDL-C. Again HDL-C level was also increased by the treatment of Glipizide when compared to the control group. A low level of HDL-C is an unsuitable feature for cardioprotection. Ginsberg and Goldberg76 found that the impact of Glipizide on HDL-C level is not undesirable.
CONCLUSION
From the results of the study, it can be concluded that Glipizide shows a comparable anxiolytic and antidepressant activity. Moreover, Glipizide treatment also significantly reduced serum blood glucose level and body weight while showing no toxic effects on the average weight of liver and kidney, however, has low effects on the abdominal and visceral fat deposition. The present study concluded that Glipizide has less suppressive effects on lipid profile which signifies that Glipizide has low organ toxicity while might have some anxiolytic effects with a warrant of further long spectrum animal and human studies.
SIGNIFICANCE STATEMENT
This study discovered the influences of glipizide on lipid index, major organs and CNS that can be beneficial for patients and clinicians to better management of T2DM. This study will help the researchers to uncover the critical pharmacological and toxicological aspects of Glipizide that many researchers were not able to explore.
ACKNOWLEDGMENT
The authors sincerely thank the pharmacy discipline, faculty of science, Noakhali Science and Technology University, Bangladesh for providing adequate financial support, laboratory facilities and necessary reagents to carry out the research work.
REFERENCES
- American Diabetes Association, 2010. Diagnosis and classification of diabetes mellitus. Diabetes Care, 33: S62-S69.
CrossRefDirect Link - Katsarou, A., S. Gudbjörnsdottir, A. Rawshani, D. Dabelea and E. Bonifacio et al., 2017. Type 1 diabetes mellitus. Nat. Rev. Dis. Primers, Vol. 3.
CrossRefDirect Link - NCD Risk Factor Collaboration (NCD-RisC), 2016. Worldwide trends in diabetes since 1980: A pooled analysis of 751 population-based studies with 4·4 million participants. Lancet, 387: 1513-1530.
CrossRefDirect Link - Saeedi, P., I. Petersohn, P. Salpea, B. Malanda and S. Karuranga et al., 2019. Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: Results from the international diabetes federation diabetes atlas, 9th edition. Diabetes Res. Clin. Pract., Vol. 157.
CrossRefDirect Link - Saquib, N., J. Saquib, T. Ahmed, M.A. Khanam and M.R. Cullen, 2012. Cardiovascular diseases and type 2 diabetes in Bangladesh: A systematic review and meta-analysis of studies between 1995 and 2010. BMC Public Health, Vol. 12.
CrossRefDirect Link - Wild, S., G. Roglic, A. Green, R. Sicree and H. King, 2004. Global prevalence of diabetes: Estimates for the year 2000 and projections for 2030. Diabetes Care, 27: 1047-1053.
CrossRefPubMedDirect Link - Akter, S., M.M. Rahman, S.K. Abe and P. Sultana, 2014. Prevalence of diabetes and prediabetes and their risk factors among Bangladeshi adults: A nationwide survey. Bull. World Health Organ., 92: 204-213A.
CrossRefDirect Link - Bystritsky, A., S.S. Khalsa, M.E. Cameron and J. Schiffman, 2013. Current diagnosis and treatment of anxiety disorders. Pharm. Ther., 38: 30-57.
Direct Link - Lustman, P.J., R.J. Anderson, K.E. Freedland, M. de Groot, R.M. Carney and R.E. Clouse, 2000. Depression and poor glycemic control: A meta-analytic review of the literature. Diabetes Care, 23: 934-942.
CrossRefDirect Link - Kan, C., N. Silva, S.H. Golden, U. Rajala, M. Timonen, D. Stahl and K. Ismail, 2013. A systematic review and meta-analysis of the association between depression and insulin resistance. Diabetes Care, 36: 480-489.
CrossRefDirect Link - Grigsby, A.B., R.J. Anderson, K.E. Freedland, R.E. Clouse and P.J. Lustman, 2002. Prevalence of anxiety in adults with diabetes: A systematic review. J. Psychosomatic Res., 53: 1053-1060.
CrossRefDirect Link - Agrawal, J., R. Kumar, P. Malhi and D. Dayal, 2016. Prevalence of psychosocial morbidity in children with type 1 diabetes mellitus: A survey from Northern India. J. Pediatr. Endocrinol. Metab., 29: 893-899.
CrossRefDirect Link - Roy, T. and C.E. Lloyd, 2012. Epidemiology of depression and diabetes: A systematic review. J. Affective Disord., 142: S8-S21.
CrossRefDirect Link - Chehade, J.M., M. Gladysz and A.D. Mooradian, 2013. Dyslipidemia in type 2 diabetes: Prevalence, pathophysiology, and management. Drugs, 73: 327-339.
CrossRefDirect Link - Chapman, M.J., H.N. Ginsberg, P. Amarenco, F. Andreotti and J. Borén et al., 2011. Triglyceride-rich lipoproteins and high-density lipoprotein cholesterol in patients at high risk of cardiovascular disease: Evidence and guidance for management. Eur. Heart J., 32: 1345-1361.
CrossRefPubMedDirect Link - Taskinen, M.R., 2002. Diabetic dyslipidemia. Atherosclerosis Suppl., 3: 47-51.
CrossRefPubMedDirect Link - Taskinen, M.R., 2003. Diabetic dyslipidaemia: From basic research to clinical practice. Diabetologia, 46: 733-749.
CrossRefDirect Link - Cartee, G.D., R.T. Hepple, M.M. Bamman and J.R. Zierath, 2016. Exercise promotes healthy aging of skeletal muscle. Cell Metab., 23: 1034-1047.
CrossRefDirect Link - Mahato, R.V., P. Gyawali, P.P. Raut, P. Regmi, K.P. Singh, D.R. Pandeya and P. Gyawali, 2011. Association between glycaemic control and serum lipid profile in type 2 diabetic patients: Glycated haemoglobin as a dual biomarker. Biomed. Res., 22: 375-380.
Direct Link - Artha, I.M.J.R., A. Bhargah, N.K. Dharmawan, U.W. Pande and K.A. Triyana et al., 2019. High level of individual lipid profile and lipid ratio as a predictive marker of poor glycemic control in type-2 diabetes mellitus. Vasc. Health Risk Manage., 15: 149-157.
CrossRefDirect Link - Benjamin, E.J., S.S. Virani, C.W. Callaway, A.M. Chamberlain and A.R. Chang et al., 2018. Heart disease and stroke statistics-2018 update: A report from the American heart association. Circulation, 137: e67-e492.
CrossRefDirect Link - Howard, B.V., D.C. Robbins, M.L. Sievers, E.T. Lee and D. Rhoades et al., 2000. LDL cholesterol as a strong predictor of coronary heart disease in diabetic individuals with insulin resistance and low LDL. Arteriosclerosis, Thrombosis, Vasc. Biol., 20: 830-835.
CrossRefDirect Link - Aronson, D. and E.R. Edelman, 2014. Coronary artery disease and diabetes mellitus. Cardiol. Clin., 32: 439-455.
CrossRefDirect Link - Schofield, J.D., Y. Liu, P. Rao-Balakrishna, R.A. Malik and H. Soran, 2016. Diabetes dyslipidemia. Diabetes Ther., 7: 203-219.
CrossRefDirect Link - Scheen, A.J., 2014. Pharmacokinetic and toxicological considerations for the treatment of diabetes in patients with liver disease. Expert Opin. Drug Metab. Toxicol., 10: 839-857.
CrossRefDirect Link - Arnouts, P., D. Bolignano, I. Nistor, H. Bilo, L. Gnudi, J. Heaf and W. van Biesen, 2014. Glucose-lowering drugs in patients with chronic kidney disease: A narrative review on pharmacokinetic properties. Nephrol. Dialysis Transplant., 29: 1284-1300.
CrossRefDirect Link - Sibeko, L. and T. Johns, 2021. Global survey of medicinal plants during lactation and postpartum recovery: Evolutionary perspectives and contemporary health implications. J. Ethnopharmacol., Vol. 270.
CrossRefDirect Link - Chen, U., M.S. Hussain, T. Mazumder, S.M.N. Uddin and S. Banik, 2019. Neuropharmacological evaluation of methanolic extract of Costus speciosus Linn. rhizome in Swiss albino mice. Asian Pac. J. Trop. Biomed., 9: 217-221.
CrossRefDirect Link - Hui, Z.G., X.W. Zhou, R.J. Li, Y.B. Wang and J. Ma, 2015. Studies on the extraction process of total flavonoids in Radix puerariae and their hypoglycemic effect in mice. Biomed. Res., 26: 51-54.
Direct Link - Miaffo, D., F. Ntchapda, T.A. Mahamad, B. Maidadi and A. Kamanyi, 2021. Hypoglycemic, antidyslipidemic and antioxydant effects of Vitellaria paradoxa barks extract on high-fat diet and streptozotocin-induced type 2 diabetes rats. Metab. Open, Vol. 9.
CrossRefDirect Link - Ahmad, F. and M.K. Richardson, 2013. Exploratory behaviour in the open field test adapted for larval zebrafish: Impact of environmental complexity. Behav. Processes, 92: 88-98.
CrossRefDirect Link - Campos, A.C., M.V. Fogaca, D.C. Aguiar and F.S. Guimaraes, 2013. Animal models of anxiety disorders and stress. Rev. Bras. Psiquiatr., 35: S101-S111.
CrossRefDirect Link - Tye, K.M., J.J. Mirzabekov, M.R. Warden, E.A. Ferenczi and H.C. Tsai et al., 2013. Dopamine neurons modulate neural encoding and expression of depression-related behaviour. Nature, 493: 537-541.
CrossRefDirect Link - Adhikari, A., M.A. Topiwala and J.A. Gordon, 2010. Synchronized activity between the ventral hippocampus and the medial prefrontal cortex during anxiety. Neuron, 65: 257-269.
CrossRefDirect Link - Cryan, J.F. and F.F. Sweeney, 2011. The age of anxiety: Role of animal models of anxiolytic action in drug discovery. Br. J. Pharmacol., 164: 1129-1161.
CrossRefDirect Link - Ye, Q., H. Lei, Z. Fan, W. Zheng and S. Zheng, 2014. Difference in LDL receptor feedback regulation in macrophages and vascular smooth muscle cells: Foam cell transformation under inflammatory stress. Inflammation, 37: 555-565.
CrossRefDirect Link - Mokha, J.S., S.R. Srinivasan, P. DasMahapatra, C. Fernandez, W. Chen, J. Xu and G.S. Berenson, 2010. Utility of waist-to-height ratio in assessing the status of central obesity and related cardiometabolic risk profile among normal weight and overweight/obese children: The Bogalusa Heart Study. BMC Pediatr., Vol. 10.
CrossRefDirect Link - Wong, N.D., 2014. Epidemiological studies of CHD and the evolution of preventive cardiology. Nat. Rev. Cardiol., 11: 276-289.
CrossRefDirect Link - Ezeja, M.I., A.O. Anaga and I.U. Asuzu, 2014. Acute and sub-chronic toxicity profile of methanol leaf extract of Gouania longipetala in rats. J. Ethnopharmacol., 151: 1155-1164.
CrossRefDirect Link - Baranwal, J.K., R. Maskey, S. Majhi, M. Lamsal and N. Bara, 2015. Association between level of HbA1c and lipid profile in T2DM patients attending diabetic OPD at BPKIHS. Health Renaissance, 13: 16-23.
CrossRefDirect Link - Kessler, R.C., P. Berglund, O. Demler, R. Jin and D. Koretz et al., 2003. The epidemiology of major depressive disorder: Results from the national comorbidity survey replication (NCS-R). JAMA, 289: 3095-3105.
CrossRefPubMedDirect Link - Whiting, D.R., L. Guariguata, C. Weil and J. Shaw, 2011. IDF diabetes atlas: Global estimates of the prevalence of diabetes for 2011 and 2030. Diabetes Res. Clin. Pract., 94: 311-321.
CrossRefDirect Link - Holt, R.I.G., 2011. Undoing descartes: Integrating diabetes care for those with mental illness. Pract. Diabetes Int., 28: 270-275.
CrossRefDirect Link - Popescu, S., B. Timar, F. Baderca, M. Simu, L. Diaconu, I. Velea and R. Timar, 2016. Age as an independent factor for the development of neuropathy in diabetic patients. Clin. Interventions Aging, 11: 313-318.
CrossRefDirect Link - Roy, T., C.E. Lloyd, M. Parvin, K.G.B. Mohiuddin and M. Rahman, 2012. Prevalence of co-morbid depression in out-patients with type 2 diabetes mellitus in Bangladesh. BMC Psychiatry, Vol. 12.
CrossRefDirect Link - de Groot, M., M. Kushnick, T. Doyle, J. Merrill, M. McGlynn, J. Shubrook and F. Schwartz, 2010. Depression among adults with diabetes: Prevalence, impact, and treatment options. Diabetes Spectr., 23: 15-18.
CrossRefDirect Link - Liu, G.X., G.Q. Cai, Y.Q. Cai, Z.J. Sheng and J. Jiang et al., 2007. Reduced anxiety and depression-like behaviors in mice lacking GABA transporter subtype 1. Neuropsychopharmacology, 32: 1531-1539.
CrossRefDirect Link - Elumalai, P. and S. Lakshmi, 2016. Biomodulators of anxiety. Int. J. Clin. Exp. Med. Sci., 2: 7-12.
CrossRefDirect Link - Luscher, B., T. Fuchs and C.L. Kilpatrick, 2011. GABBA receptor trafficking-mediated plasticity of inhibitory synapses. Neuron, 70: 385-409.
CrossRefDirect Link - Ali, M.S., P.R. Dash and M. Nasrin, 2015. Study of sedative activity of different extracts of Kaempferia galanga in Swiss albino mice. BMC Complementary Altern. Med., Vol. 15.
CrossRefDirect Link - Okoli, C.O., A.C. Ezike, O.C. Agwagah and P.A. Akah, 2010. Anticonvulsant and anxiolytic evaluation of leaf extracts of ocimum gratissimum, a culinary herb. Pharmacogn. Res., 2: 36-40.
CrossRefDirect Link - Webster, J.P., M. Kaushik, G.C. Bristow and G.A. McConkey, 2013. Toxoplasma gondii infection, from predation to schizophrenia: Can animal behaviour help us understand human behaviour? J. Exp. Biol., 216: 99-112.
CrossRefDirect Link - McIlwain, K.L., M.Y. Merriweather, L.A. Yuva-Paylor and R. Paylor, 2001. The use of behavioral test batteries: Effects of training history. Physiol. Behav., 73: 705-717.
CrossRefPubMedDirect Link - Walf, A.A. and C.A. Frye, 2007. The use of the elevated plus maze as an assay of anxiety-related behavior in rodents. Nat. Protocols, 2: 322-328.
CrossRefDirect Link - Bahramsoltani, R., M.H. Farzaei, M.S. Farahani and R. Rahimi, 2015. Phytochemical constituents as future antidepressants: A comprehensive review. Rev. Neurosci., 26: 699-719.
CrossRefDirect Link - Murai, Y., T. Ohta, H. Tadaki, K. Miyajima, M. Shinohara, F. Fatchiyah and T. Yamada, 2017. Assessment of pharmacological responses to an anti-diabetic drug in a new obese type 2 diabetic rat model. Med. Arch., 71: 380-384.
CrossRefDirect Link - Anderson, M., J. Powell, K.M. Campbell and J.R. Taylor, 2014. Optimal management of type 2 diabetes in patients with increased risk of hypoglycemia. Diabetes Metab. Syndr. Obesity: Targets Ther., 7: 85-94.
CrossRefDirect Link - Sommerfield, A.J., I.J. Deary and B.M. Frier, 2004. Acute hyperglycemia alters mood state and impairs cognitive performance in people with type 2 diabetes. Diabetes Care, 27: 2335-2340.
CrossRefDirect Link - Ridderstråle, M., S. Gudbjörnsdottir, B. Eliasson, P.M. Nilsson and J. Cederholm, 2006. Obesity and cardiovascular risk factors in type 2 diabetes: Results from the Swedish National Diabetes Register. J. Int. Med., 259: 314-322.
CrossRefDirect Link - Cefalu, W.T., J.B. Buse, S. del Prato, P.D. Home and D. LeRoith et al., 2014. Beyond metformin: Safety considerations in the decision-making process for selecting a second medication for type 2 diabetes management: Reflections from a diabetes care editors’ expert forum. Diabetes care, 37: 2647-2659.
CrossRefDirect Link - Malik, M.Y., S. Jaiswal, A. Sharma, M. Shukla and J. Lal, 2016. Role of enterohepatic recirculation in drug disposition: Cooperation and complications. Drug Metab. Rev., 48: 281-327.
CrossRefDirect Link - Qaseem, A., L.L. Humphrey, D.E. Sweet, M. Starkey and P. Shekelle, 2012. Oral pharmacologic treatment of type 2 diabetes mellitus: A clinical practice guideline from the American College of Physicians. Ann. Internal Med., 156: 218-231.
CrossRefDirect Link - Kahn, S.E., S.M. Haffner, M.A. Heise, W.H. Herman and R.R. Holman et al., 2006. Glycemic durability of rosiglitazone, metformin, or glyburide monotherapy. N. Engl. J. Med., 355: 2427-2443.
CrossRefDirect Link - Wang, H., Y. Ni, S. Yang, H. Li, X. Li and B. Feng, 2013. The effects of gliclazide, metformin, and acarbose on body composition in patients with newly diagnosed type 2 diabetes mellitus. Curr. Ther. Res., 75: 88-92.
CrossRefDirect Link - Krauss, R.M., 2004. Lipids and lipoproteins in patients with type 2 diabetes. Diabetes Care, 27: 1496-1504.
CrossRefDirect Link - Wu, L. and K.G. Parhofer, 2014. Diabetic dyslipidemia. Metab.: Clin. Exp., 63: 1469-1479.
CrossRefDirect Link - Goldberg, I.J., 2001. Diabetic dyslipidemia: Causes and consequences. J. Clin. Endocrinol. Metab., 86: 965-971.
CrossRefPubMedDirect Link - Santamarina-Fojo, S., G. Lambert, J.M. Hoeg and H.B. Brewer Jr., 2000. Lecithin-cholesterol acyltransferase: Role in lipoprotein metabolism, reverse cholesterol transport and atherosclerosis. Curr. Opin. Lipidology, 11: 267-275.
CrossRefDirect Link - Guimarães, P.R., A.M.P. Galvão, C.M. Batista, G.S. Azevedo and R.D. Oliveira et al., 2000. Eggplant (Solanum melongena) infusion has a modest and transitory effect on hypercholesterolemic subjects. Braz. J. Med. Biol. Res., 33: 1027-1036.
CrossRefDirect Link - Akpan, E.J., J.E. Okokon and E. Offong, 2012. Antidiabetic and hypolipidemic activities of ethanolic leaf extract and fractions of Melanthera scandens. Asian Pac. J. Trop. Biomed., 2: 523-527.
CrossRefDirect Link - Rajaei, Z., M.A.R. Hadjzadeh, R. Moradi, A. Ghorbani and A. Saghebi, 2015. Antihyperglycemic and antihyperlipidemic effects of hydroalcoholic extract of Securigera securidaca seeds in streptozotocin-induced diabetic rats. Adv. Biomed. Res., Vol. 4.
CrossRefDirect Link - Murali, B., U.M. Upadhyaya and R.K. Goyal, 2002. Effect of chronic treatment with Enicostemma littorale in non-insulin-dependent diabetic (NIDDM) rats. J. Ethnopharmacol., 81: 199-204.
CrossRefPubMedDirect Link - Stancu, C. and A. Sima, 2001. Statins: Mechanism of action and effects. J. Cell. Mol. Med., 5: 378-387.
CrossRefDirect Link - Emerging Risk Factors Collaboration, 2010. Diabetes mellitus, fasting blood glucose concentration and risk of vascular disease: A collaborative meta-analysis of 102 prospective studies. Lancet, 375: 2215-2222.
CrossRefPubMedDirect Link - Abba, A.B. and D.A. Abbas, 2021. Evaluation of lipid profile and inflammatory parameters in female diabetes type 2 induced rabbits treated with glimepride, bromocriptine and fluoxtein. Iraqi J. Vet. Med., 42: 97-104.
CrossRefDirect Link - Ginsberg, H.N. and I.J. Goldberg, 2011. The pancreas and lipoprotein metabolism. Compr. Physiol.
CrossRefDirect Link