ABSTRACT
Background and Objective: Biofertilizers are cost-effective and eco-friendly while chemicals fertilizer are not only costly but also pollutes our natural resources. The objective of this study was to search out a better combination of PGPR with chemical fertilizer that can reduce the inorganic fertilizer requirement for wheat crops. Materials and Methods: This pot study was carried out in a greenhouse of NARC Islamabad in a completely randomized design. Three nitrogen levels, Zero (ON), 75% (75 N) and 100% (100 N)of recommended dose (120kg/N/ha) and three PGPR, WPR-32 (Azotobacter), WPR-42 (Azospirillum) and WPR-51 (Azospirillum) were used in four treatments (T1 = No inoculum, T2 = WPR-32, T3 = WPR-42, T4 = WPR-51 and T5 = WPR-32+42+51). Results: The results of all treatments showed improvement in various growth and yield parameters of wheat (Shoot Length (cm), Shoot dry weight (g/plant), Root dry weight (g plant), Grain Yield (g/plant), Straw yield (g/plant), Grain nitrogen (% plant), Grain phosphorus (%), Grain protein contents (% plant). The combined application of isolates (WPR-32+WPR-42+WPR-51) gave 41%, 27% and 4.68%, respectively higher yield over control at three levels of fertilizer. Whereas, the single application of isolates gave an average increase of 18% and 24%, respectively over control at two levels of fertilizer (0 N, 75 N). While, an increase of 16% was recorded over control at 100 N. The combined inoculation of isolates at 75% (75 N) gave the grain yield at par to 100 N application. Conclusion: It was concluded that nitrogen fertilizer application can be reduced by at least 25% without compromising on the yield of the wheat crop. The PGPR application is economical for the farmer and also environmentally friendly as well.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/jas.2023.120.131
URL: https://scialert.net/abstract/?doi=jas.2023.120.131
INTRODUCTION
Wheat (Triticum aestivum L.) is grown in a major portion of the world’s cultivated area. This crop is harvested somewhere in the world every month of the year. In most areas of the world, it is the principal food of man. At the world level, 80% of wheat production comes from these countries namely Australia, China, Canada, European Union, Pakistan, India, Turkey, Russia, the United States and Ukraine. Pakistan contributes 3.17% of world wheat production and is the 8th largest wheat producer country. The wide use of synthetic fertilizers and pesticides on crops and vegetables are currently under discussion because of environmental pollution and fear for human health. One of the reason for static crop yields is inadequate and imbalanced use of fertilizers and the country faces challenges to improve crop productivity per unit of land. Biofertilizers consist of different strains of soil microorganisms. These microbes through biological processes convert the unavailable form of an element into an available form which is nutritionally important for plants1. Microorganisms improve root systems asa result in more uptake of water and nutrients from the soil. The beneficial plant-microbes interactions in the rhizosphere are determinants of plant health, soil fertility and recycling of nutrients2,3.The use of biofertilizers has several benefits over inorganic chemicals for agricultural purposes: (i) These fertilizers are more environment friendly than many of the in-use conventional chemicals, (ii) Neither noxious matters from microorganisms nor themselves add to the food chain and (iii) Self and rapid multiplication of microbe avoids the need forcontinualapplications4,5.
The objective of this study was to search out a better combination of PGPR with chemical fertilizer that can reduce the inorganic fertilizer requirement for the wheat crops.
MATERIALS AND METHODS
This study was carried out during 2013-2014 in greenhouse of National Agriculture Research Center, Islamabad-Pakistan and designed to record the response of wheat (Triticum aestivum L.) to different combinations of PGPR strains at three levels of nitrogen 0, 75% (75 N), 100% (100 N) of the recommended dose (120 kg/N/ha). The microorganisms used in this study was obtained from the Soil Biology and Biochemistry Laboratory of Land Resources Research Institute of NARC. Single super phosphate (SSP) and Sulphate of Potash (K2SO4) were applied as P and K sources whereas urea was used as N-source. The experiment was laid out using a two-factor factorial completely randomized design with three replications. The wheat variety (GA-2002) was sown in pots having 8 kg sterilized soil per pot.
Data were recorded at the harvesting stage. Sterilized soil was used in the pots to avoid contamination from any indigenous microorganisms. Soil analysis was done before filling pots. Three best PGPR isolates were selected based their nitrogenase ability and phytohormone production i.e., WPR-32 (Azotobacter), WPR-42 (Azospirillum) and WPR-51 (Azospirillum).These bacterial strains were cultured in LB broth in 250 mL flasks. These were incubated for 2-3 days (according to requirement). The total numbers of cells in each inoculum were 10×10–8, maintained by optical density at 550 nm. These cultures were shifted to the carrier (soil rich inorganic matter) of biofertilizer at NARC. Wheat seeds were coated with slurry, which was prepared by mixing 10% sugar solution in a biofertilizer packet. The combinations of PGPR with carrier were made as desired in treatments. Control was treated with carrier alone. Six seeds were sown per pot at a depth of 1.3cm. Plant to plant distance was maintained by thinning and four seedlings were kept in each pot. Fertilizer was applied at sowing, phosphorus and potassium were used in a single dose whereas nitrogen was used in two split doses, first at sowing and second after 30 days. There were five treatments including control, 03 N rates and three replications. Altogether, 30 pots were used in the study. After germination, plants were irrigated with tap water when required.
| Treatments: | ||
| T1 | = | (Ctrl) No inoculum |
| T2 | = | WPR-32 (Azotobacter) |
| T3 | = | WPR-42 (Azospirillum) |
| T4 | = | WPR-51 (Azospirillum) |
| T5 | = | PR-32+42+51 Azotobacter and Azospirillum |
Seed phosphorus content (%): The finely ground seed sample (0.25 g), was taken in a conical flask and 10 mL mixed acid (perchloric and nitric acid in 2:1) was added. These flasks were heated on a hot plate at a temperature of 100-300°C until the solution samples (about 2-3 mL) were colourless. Flasks were removed from hot plates and cooled. The digest was transferred into a 50 mL volume tric flask and a volume (50 mL) was made with distilled water6. For P estimation, 5 mL extract was taken in a test tube and 5 mL color-developing reagent (ammonium molybdate+ammonium vanadate+nitric acid) was added. The tubes were stirred in a mechanical stirrer, Hamilton Beach (Scovill), Virginia USA.
Then 25 mL boric acid (4%) solution was transferred to a 250 mL receiver flask and distillation was done. After 3 min when distillation was completed digestion tube was removed and the sample was titrated with 0.1 N HCl. The volume of HCl used was recorded5 and nitrogen was calculated by the following formula:
N (%) = 14.0067× (mL of titrant for sample-mL of titrant for blank)×Normality of acid weight of sample×10 |
Seed nitrogen content (%): Dry ground seed samples (0.5 g) were transferred in digestion tubes One Kjeldahl tablet and 10 mL conc. H2SO4 was added to the tubes. These tubes were placed in digester at 420°C under a fume hood (HEMCO Corporation, 711 South Powell Road, Independence, USA). After 1 hr these tubes were removed from the digester (Hot plate, Barnstead, Thomas Scientific P.O. Box 99, Swedesboro, NJ 08085) was taken in a test tube and 5 mL color developing reagent (ammonium molybdate+ammonium vanadate+ nitric acid) was added. The tubes were stirred in a mechanical stirrer (Hamilton Beach (Scovill), Virginia USA) carefully and cooled. Distilled water (75 mL) was added to each digestion tube and mixed well.
Plant sampling: Seed samples were taken at the harvest stage for nitrogen and phosphorus content from each pot. The readings were taken on a spectrophotometer, (PG Instruments T60 UV/VIS spectrophotometer, Wood way Lane, Alma Park, Leicestershire LE17 5FB, United Kingdom) at 882 nm7.
RESULTS
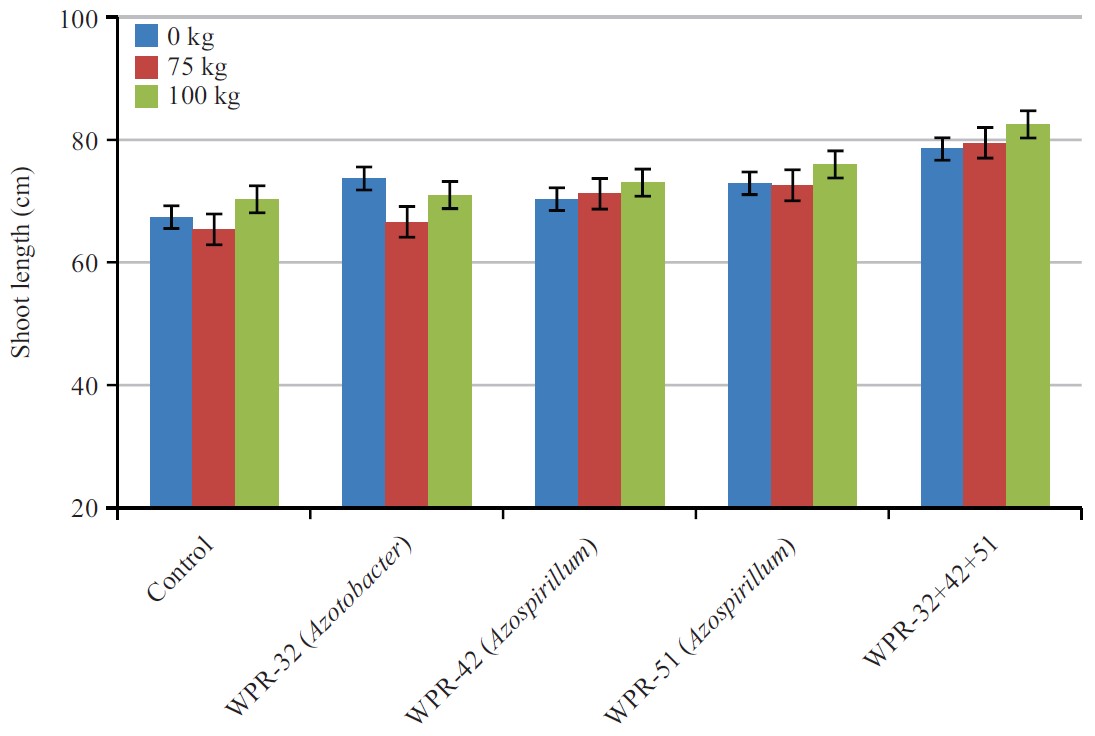
Shoot length (cm): The effect of PGPR, N fertilization and the interaction of inoculants and fertilizers on shootlength were presented in Fig. 1. By taking the average values of different N treatments including control, the calculated values showed the mean effect of strains on shoot length. Results indicated that PGPR had a significant effect on shoot length. In the control i.e., soil without PGPR inoculation, shoot length was 66.2 cm, which increased significantly to 73.7 cm following the application of WPR-32, WPR-42, WPR-51 and a mixture of these three PGPR. The PGPR strains, WPR-32, WPR-42 and WPR-51 alone did not show any significant increase in plant height. The maximum shoot length (79.1 cm) was recorded in a mixture of three PGPR, which was 9.9% higher than the height of the control. Effect of two N rates on plant height was also significant (p<0.05). The average shootlength in unfertilized control was 69.7 cm. Application of N at the rates of 75 and 100 kg/N/ha significantly increased the height to 73 and 75 cm, respectively. The difference between the two N rates was also significant. By taking the average of two N rates, the height of the plant was 74 cm as compared to 69 cm in unfertilized soil showing a 6% increase over run fertilized soil. The interactive effect of the isolates and N rates showed significantly higher shoot length relative to the values recorded for individual treatments of either isolates or N rates. The maximum shoot length (80 cm) was recorded for a mixture of WPR-32, WPR-42, WPR-51 at 100 N, which was statistically at par with the mixture treatment at 75 N.
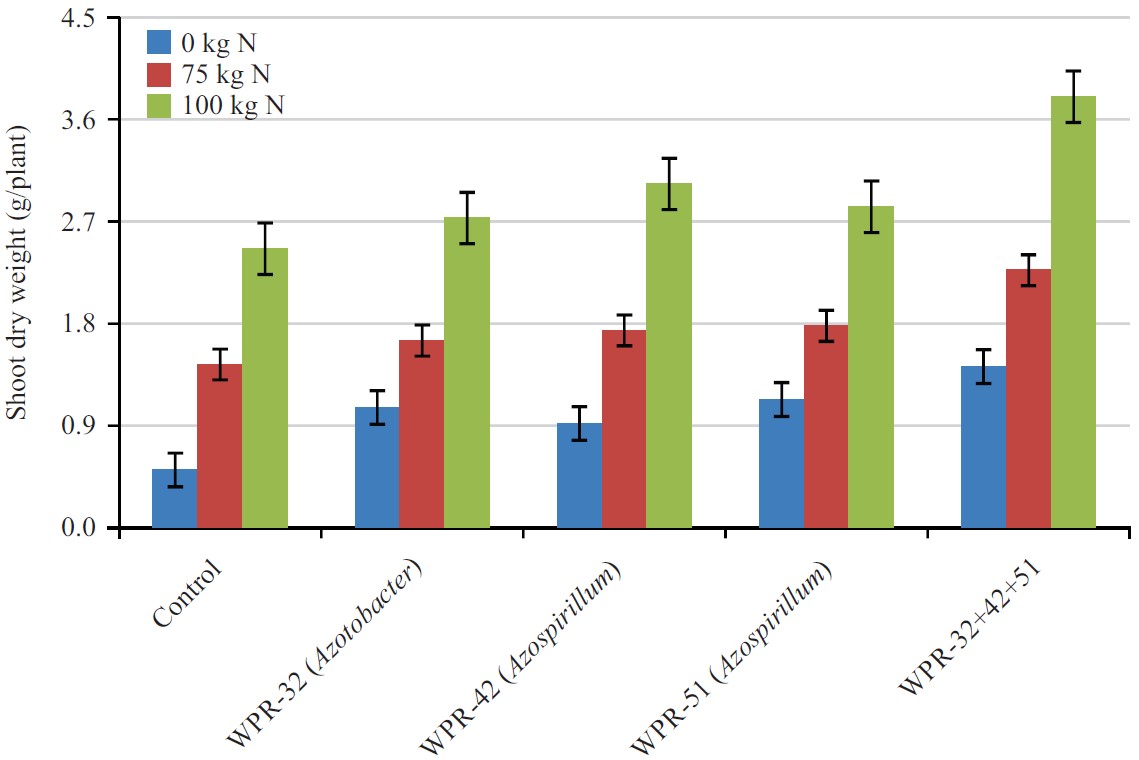
Shoot dry weight (g/plant): Effect of PGPR, N fertilization and the interaction of inoculants and fertilizers on shoot dry weight were presented in Fig. 2. By taking the average values of different N treatments including control, the calculated values showed the mean effect of strains on shoot dry weight. Results indicated that PGPR had significant effect on shoot dry weight.
| Fig. 1: | Combine effect of nitrogen fertilizer and PGPR on wheat shoot length |
| Fig. 2: | Combine effect of nitrogen fertilizer and PGPR on wheat shoot dry weight |
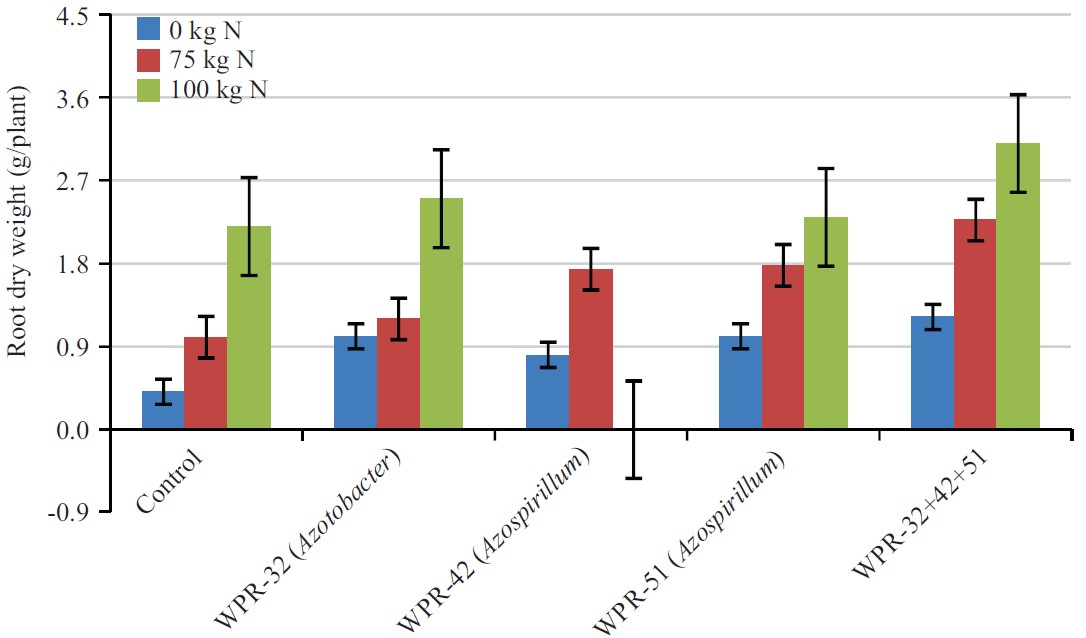
| Fig. 3: | Combine Effect of nitrogen fertilizer and PGPR on root dry weight of wheat |
In the control soil without PGPR inoculation, shoot dry weight was 1.47 g, which increased significantly to 2.49 g following the application of WPR-32, WPR-42, WPR-51 and mixture of these three PGPR. The PGPR strains, WPR-32, WPR-42 and WPR-51 alone did not show any significant increase in shoot dry weight. The maximum shoot dry weight (2.49 g) was recorded in mixture of three PGPR, which was 69.3% higher than the weight of the control. Effect of two N rates on shoot dry weight was also significant (p<0.05). The average shoot dry weight in unfertilized control was 1.04 g. Application of N at the rates of 75 and 100 kg/N/ha significantly increased the shoot dry weight to 1.82 and 3.10 g, respectively. The difference between the two N rates was also significant. By taking the average of two N rates, the height of the plant was 2.46 g as compared to 1.47 g in unfertilized soil showing 67.3% increase over unfertilized soil. The interactive effect of the isolates and N rates showed significantly higher shoot dry weight relative to the values recorded for individual treatments of either isolates or N rates. The maximum shoot dry weight (3.80 g) was recorded for mixture of WPR-32, WPR-42, WPR-51 at 100 N.
Root dry weight (g/plant): Effect of PGPR, N fertilization and the interaction of inoculants and fertilizers on root dry weight presented in Fig. 3. By taking the average values of different N treatments including control, the calculated values showed the mean effect of strains on root dry weight.
Results indicated that PGPR had significant effect on root dry weight. In the control soil without PGPR inoculation, root dry weight was 66.2 cm, which increased significantly to 73.7 cm following the application of WPR-32, WPR-42, WPR-51 and mixture of these three PGPR.
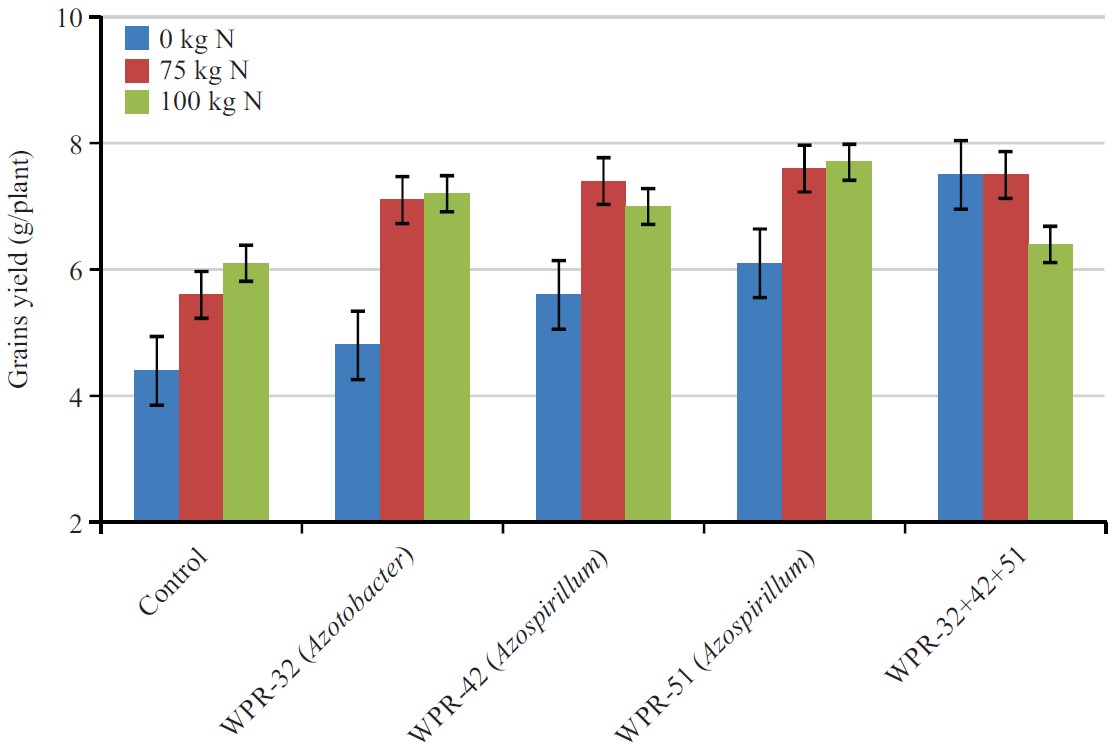
| Fig. 4: | Combine effect of nitrogen fertilizer and PGPR on wheat grain yield (g/plant) |
The PGPR strains, WPR-32, WPR-42 and WPR-51alone did not show any significant increase in root dry weight. The maximum root dry weight (79.1 cm) was recorded in mixture of three PGPR, which was 9.9% higher than the root dry weight of the control. Effect of two N rates on root dry weight was also significant (p<0.05). The average root dry weight in unfertilized control was 69.7 cm. Application of N at the rates of 75 and 100 kg/N/ha significantly increased the weight to 73 and 75 cm, respectively. The difference between the two N rates was also significant. By taking the average of two N rates, the root dry weight was 74 cm as compared to 69 cm in unfertilized soil showing a 6% increase over unfertilized soil. The interactive effect of the isolates and N rates showed significantly higher root dry weight relative to the values recorded for individual treatments of either isolates or N rates. The maximum root dry weight (75 cm) was recorded for a mixture of WPR-32, WPR-42, WPR-51 at 100 N, which was statistically at par with mixture treatment at 75 N.
Grain yield (g/plant): Effect of PGPR, N fertilization and the interaction of inoculants and fertilizers on grain yield were presented in Fig. 4. By taking the average values of different N treatments including control, the calculated values showed the mean effect of strains on grain yield.
Results indicated that PGPR had a significant effect on grain yield. In the control soil without PGPR inoculation, grain yield was 5.4 g, which increased significantly to 7.4 g following the application of WPR-32, WPR-42, WPR-51 and a mixture of these three PGPR. The PGPR strains, WPR-32, WPR-42 and WPR-51alone also show significant increase in grain yield.
The maximum grain yield (7.4 g) was recorded in mixture of three PGPR, which was 37% higher than the grain yield of the control. Effect of two N rates on grain yield was also significant (p<0.05). The average grain yield in unfertilized control was 5.6 g. Application of N at the rates of 75 and 100 kg/N/ha significantly increased the grain yield to 7.1 and 7.3 g, respectively. The difference between the two N rates was also significant. By taking the average of two N rates, the grain yield was7.2 g as compared to 5.6 g in unfertilized soil showing 28.5% increase over unfertilized soil.
The interactive effect of the isolates and N rates showed significantly higher grain yield relative to the values recorded for individual treatments of either isolates or N rates. The maximum grain yield (7.5 g) was recorded in mixture of WPR-32, WPR-42, WPR-51 at 75 N.
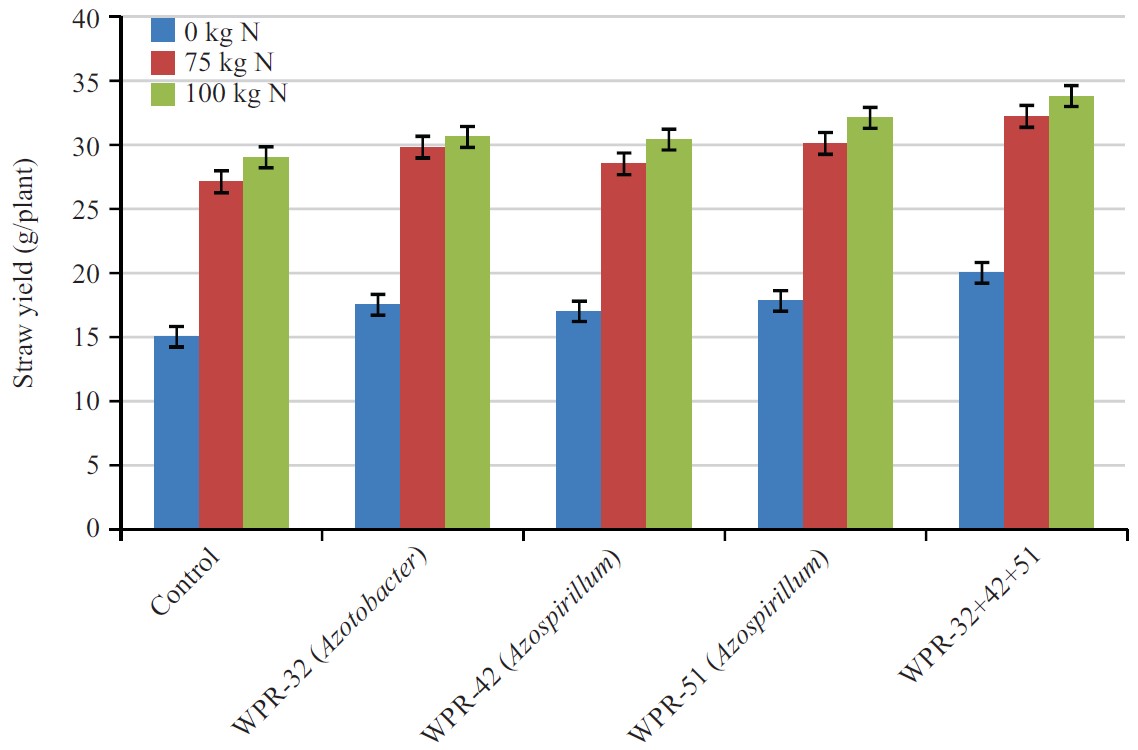
Straw yield (g/plant): Effect of PGPR, N fertilization and the interaction of inoculants and fertilizers on straw yield were presented in Fig. 5. By taking the average values of different N treatments including control, the calculated values showed the mean effect of strains on straw yield. Results indicated that PGPR had significant effect on straw yield. In the control soil without PGPR inoculation, straw yield was 23.7 g, which increased significantly to 28.6 g following the application of WPR-32, WPR-42, WPR-51 and mixture of these three PGPR.
The PGPR strains, WPR-32, WPR-42 and WPR-51 alone did not show any significant increase in straw yield. The maximum straw yield (28.6 g) was recorded in mixture of three PGPR, which was 20.6% higher than the straw yield of the control.
Effect of two N rates on straw yield was also significant (p<0.05). The average straw yield in unfertilized control was 17.5 g. Application of N at the rates of 75 and 100 kg/N/ha significantly increased the straw yield to 30.1 and 31.8 g, respectively.
| Fig. 5: | Combine effect of nitrogen fertilizer and PGPR on wheat straw yield) g/plant |
| Table1: | Effect of PGRR and nitrogen rates on grain protein, phosphorus and protein content (of wheat) | ||
| Nitrogen levels (120 kg ha–1) | |||
| Treatments | 0 N | 75 N | 100 N |
| Grain nitrogen (% plant) | |||
| Control (without inoculation) | 1.24i | 1.44d | 1.27gh |
| WPR-32 (Azotobacter) | 1.29gh | 1.54bc | 1.33efg |
| WPR-42 (Azospirillum) | 1.32fg | 1.56bc | 1.48c |
| WPR-51 (Azospirillum) | 1.38ef | 1.44de | 1.55bc |
| WPR-32+42+51 | 1.5bc | 1.72a* | 1.68a |
| Grain phosphorous (% plant) | |||
| Control (without inoculation) | 0.22jk | 0.35ghi | 0.48de |
| WPR-32 (Azotobacter) | 0.31hij | 0.42ef | 0.55cd |
| WPR-42 (Azospirillum) | 0.30hij | 0.45e | 0.54cd |
| WPR-51 (Azospirillum) | 0.38fgh | 0.49de | 0.58ab |
| WPR-32+42+51 | 0.41efg | 0.52bc | 0.62a* |
| Grain protein contents (% plant) | |||
| Control (without inoculation) | 7.25i | 9.0d | 7.93gh |
| WPR-32 (Azotobacter) | 8.06gh | 9.62bc | 8.31efg |
| WPR-42 (Azospirillum) | 8.25fg | 9.75bc | 9.25c |
| WPR-51 (Azospirillum) | 8.31efg | 9.0de | 10.5ab |
| WPR-32+42+51 | 8.87def | 10.7a* | 9.68bc |
| Every entry in the table is the mean of three repeats and values with *Means significantly (p<0.05) different | |||
The difference between the two N rates was also significant. By taking the average of two N rates, the straw yield was 30.6 g as compared to 17.5 g in unfertilized soil showing 74.8 % increase over unfertilized soil. The interactive effect of the isolates and N rates showed significantly higher straw yield relative to the values recorded for individual treatments of either isolates or N rates. The maximum straw yield (33.8 g) was recorded for mixture of WPR-32, WPR-42 and WPR-51 at 100 N, which was statistically at par with mixture treatment at 75 N.
Grain Nitrogen (% plant): Effect of PGPR, N fertilization and the interaction of inoculants and fertilizers on grain nitrogen was presented in Table 1. By taking the average values of different N treatments including control, the calculated values showed the mean effect of strains on grain nitrogen.
Results indicated that PGPR had significant effect on grain nitrogen. In the control soil without PGPR inoculation, grain nitrogen was 1.3%, which increased significantly to 1.6% following the application of WPR-32, WPR-42, WPR-51 and mixture of these three PGPR.
The PGPR strains, WPR-32, WPR-42 and WPR-51 alone did not show any significant increase in grain nitrogen. The maximum grain nitrogen (1.6%) was recorded in mixture of three PGPR, which was 23% higher than the height of the control.
Effect of two N rates on grain nitrogen was also significant (p<0.05). The average grain nitrogen in unfertilized control was 1.32%. Application of N at the rates of 75 and 100 kg/N/ha significantly increased the grain nitrogen to 1.56 and 1.47%, respectively. The difference between the two N rates was also significant. By taking the average of two N rates, the grain nitrogen was 1.5% as compared to1.32% in unfertilized soil showing 13.6% increase over unfertilized soil. The interactive effect of the isolates and N rates showed significantly higher grain nitrogen relative to the values recorded for individual treatments of either isolates or N rates. The maximum grain nitrogen (9.75%) was recorded for mixture of WPR-32, WPR-42, WPR-51at 75 N.
Grain phosphorus (%): Effect of PGPR, N fertilization and the interaction of inoculants and fertilizers on grain phosphorus presented in Table 1. By taking the average values of different N treatments including control, the calculated values showed the mean effect of strains on grain phosphorus. Results indicated that PGPR had significant effect on grain phosphorus. In the control soil without PGPR inoculation, grain phosphorus was 0.35%, which increased significantly to 0.51 % following the application of WPR-32, WPR-42, WPR-51 and mixture of these three PGPR. The PGPR strains, WPR-32, WPR-42 and WPR-51 alone did not show any significant increase in grain phosphorus. The maximum grain phosphorus (0.51%) was recorded in mixture of three PGPR, which was 45.7% higher than the grain phosphorus of the control. Effect of two N rates on grain phosphorus was also significant (p<0.05).
The average grain phosphorus in unfertilized control was 0.34%. Application of N at the rates of 75 and 100 kg/N/ha significantly increased the grain phosphorus to 0.45 and 0.56%, respectively. The difference between the two N rates was also significant. By taking the average of two N rates, the grain phosphorus was 0.53% as compared to 0.34% in unfertilized soil showing 55% increase over unfertilized soil. The interactive effect of the isolates and N rates showed significantly higher grain phosphorus relative to the values recorded for individual treatments of either isolates or N rates. The maximum grain phosphorus (0.62%) was recorded for mixture of WPR-32, WPR-42, WPR-51 at 100 N.
Grain protein contents (% plant): Effect of PGPR, N fertilization and the interaction of inoculants and fertilizers on grain protein presented in Table 1. By taking the average values of different N treatments including control, the calculated values showed the mean effect of strains on grain protein. Results indicated that PGPR had significant effect on grain protein. In the control soil without PGPR inoculation, grain protein was 8.06% plant, which increased significantly to 9.75% following the application of WPR-32, WPR-42, WPR-51 and mixture of these three PGPR. The PGPR strains, WPR-32, WPR-42 and WPR-51 alone did not show any significant increase in grain protein. The maximum grain protein (9.75%) was recorded in mixture of three PGPR, which was 20.9% higher than the grain protein of the control. Effect of two N rates on grain protein was also significant (p<0.05). The average grain protein in the unfertilized control was 8.37%. Application of N at the rates of 75 and 100 kg/N/ha significantly increased the grain protein to 9.76 and 9.34%, respectively. The difference between the two N rates was also significant. By taking the average of two N rates, the grain protein/plant was 9.55% as compared to 8.37% in unfertilized soil showing 18% increase over unfertilized soil. The interactive effect of the isolates and N rates showed significantly higher grain protein relative to the values recorded for individual treatments of either isolates or N rates. The maximum grain protein (10.8%) was recorded for mixture of WPR-32, WPR-51 at 75 N, which was statistically at par with mixture treatment at 100 N.
Soil nitrogen (mg kg–1): Effect of PGPR, N fertilization and the interaction of inoculants and fertilizers on soil nitrogen are were presented in Table 2. By taking the average values of different N treatments including control, the calculated values showed the mean effect of strains on soil nitrogen. Results indicated that PGPR had significant effect on soil nitrogen. In the control soil without PGPR inoculation, soil nitrogen was 0.09 mg kg–1, which decreased significantly to 0.08 mg kg–1 following the application of WPR-32, WPR-42, WPR-51 and mixture of these three PGPR. The minimum soil nitrogen (0.08 mg kg–1) was recorded in mixture of three PGPR, which was 1.25% less than the control. Effect of two N rates on soil nitrogen was also significant (p<0.05). The average soil nitrogen in unfertilized control was 0.07 mg kg–1. Application of N at the rates of 75 and 100 kg/N/ha significantly increased the soil nitrogen to 0.08 and 0.82 mg kg–1, respectively. The difference between the two N rates was also significant. By taking the average of two N rates, the soil nitrogen was 0.081 as compared to 0.07 mg kg–1 in unfertilized soil showing 15.7% decrease over unfertilized soil. The interactive effect of the isolates and N rates showed significantly lesser soil nitrogen relative to the values recorded for individual treatments of either isolates or N rates.
| Table 2: | Effect of PGRR and nitrogen rates on soil nitrogen, phosphorus, potassium (mg kg–1) of wheat | ||
| Nitrogen levels (120 kg ha–1) | |||
| Treatments | 0 N | 75 N | 100 N |
| Soil nitrogen (% plant) | |||
| Control (without inoculation) | 0.093ab | 0.097a | 0.098a |
| WPR-32 (Azotobacter) | 0.061fghi | 0.082de | 0.095ab |
| WPR-42 (Azospirillum) | 0.083cde | 0.088cd | 0.076efg |
| WPR-51 (Azospirillum) | 0.064fghi | 0.073fgh | 0.070fgh |
| WPR-32+42+51 | 0.058fghij | 0.092bc | 0.080de |
| Soil phosphorous (mg kg–1) | |||
| Control (without inoculation) | 2.8fgh | 4.5b | 6.3a |
| WPR-32 (Azotobacter) | 2.5ghi | 3.8cde | 4.8b |
| WPR-42 (Azospirillum) | 2.6fgh | 3.4def | 4.2bc |
| WPR-51 (Azospirillum) | 2.2hi | 3.0efg | 4.1bc |
| WPR-32+42+51 | 2.0ij* | 2.6fgh | 3.2efg |
| Soil potassium contents ( mg kg–1) | |||
| Control (without inoculation) | 28.0ab | 30.0a | 30.1a |
| WPR-32 (Azotobacter) | 25.6def | 27.1bc | 28.1ab |
| WPR-42 (Azospirillum) | 26.2cde | 26.8cde | 27.8bc |
| WPR-51 (Azospirillum) | 24.8defg | 24.5defg | 26.5cde |
| WPR-32+42+51 | 20.8kl | 22.5ghij | 24.2defg |
| Every entry in the table is the mean of three repeats and values with *Means significantly (p<0.05) different | |||
The minimum soil nitrogen (0.07 mg kg–1) was recorded in WPR-51 treatment at 100 N.
Soil phosphorus (mg kg–1): Effect of PGPR, N fertilization and the interaction of inoculants and fertilizers on soil phosphorus were presented in Table 2. By taking the average values of different N treatments including control, the calculated values showed the mean effect of strains on soil phosphorus.
Results indicated that PGPR had significant effect on soil phosphorus. In the control soil without PGPR inoculation, soil phosphorus was 4.5 mg kg–1, which decreased significantly to 2.6 mg kg–1 following the application of WPR-32, WPR-42, WPR-51 and mixture of these three PGPR. The minimum soil phosphorus (2.6 mg kg–1) was recorded in mixture of three PGPR, which was 73% less than the soil phosphorus of the control.
Effect of two N rates on soil phosphorus was also significant (p<0.05). The average soil phosphorus in unfertilized control was 2.3 mg kg–1. Application of N at the rates of 75 and 100 kg/N/ha significantly increased the soil phosphorus to 2.49 and 4.1 mg kg–1, respectively. The difference between the two N rates was also significant. By taking the average of two N rates, the soil phosphorus was 3.28 as compared to 2.3 mg kg–1 in unfertilized soil showing 42.6% decrease over unfertilized soil.
The interactive effect of the isolates and N rates showed significantly lesser soil phosphorus relative to the values recorded for individual treatments of either isolates or N rates.
The minimum soil phosphorus (2.0 mg kg–1) was recorded in mixture of WPR-32, WPR-42 and WPR-51 treatment at 0 N.
Soil potassium (mg kg–1): Effect of PGPR, N fertilization and the interaction of inoculants and fertilizers on soil potassium were presented in Table 2. By taking the average values of different N treatments including control, the calculated values showed the mean effect of strains on soil potassium.
Results indicated that PGPR had significant effect on soil potassium. In the control soil without PGPR inoculation, soil potassium was 29.3 mg kg–1, which decreased significantly to 24.3 mg kg–1 following the application of WPR-32, WPR-42, WPR-51 and mixture of these three PGPR. The minimum soil potassium (24.3 mg kg–1) was recorded in mixture of three PGPR, which was 20% less than the soil potassium of the control.
Effect of two N rates on soil potassium was also significant (p<0.05). The average soil potassium in unfertilized control was 23.8 mg kg–1. Application of N at the rates of 75 and 100 kg/N/ha significantly increased the soil potassium to 24.9 and 26.8 mg kg–1, respectively. The difference between the two N rates was also significant. By taking the average of two N rates, the soil potassium was 25.6 as compared to 23.8 mg kg–1 in unfertilized soil showing 7.5% decrease over unfertilized soil.
The interactive effect of the isolates and N rates showed significantly lesser soil potassium relative to the values recorded for individual treatments of either isolates or N rates. The minimum soil potassium (20.8 mg kg–1) was recorded in mixture of WPR-32, WPR-42 and WPR-51 treatment at 0 N.
DISCUSSION
In last two decades much work has been done on soil microbes to use them as bio fertilizer. This investigation was also design to cut down the fertilizer use without compromising yield. For this purpose, three different levels of nitrogen fertilizer along promising PGPR strains were used. It was found that 75 kg/N/ha along with bacterial inoculation gave yield at par to 100 kg/N/ha. Wheat (Triticum aestivum L.) is mostly cultivated crop at world level and also a main cereal crop of Pakistan. Increase in wheat production by using environment friendly technology is need of time. Farmers want to increase production of wheat per hectare with low input of chemical fertilizer. Research study showed that in case of higher application of fertilizer plants become less efficient at taking up nutrients and on other hand loss of nitrogen increases in the form of ammonia evaporation or through process of denitrification8.
Plant growth promoting rhizobacteria promote plant growth by providing growth regulators and by facilitating the uptake of nutrients from the soil9,10. The finding of this study were also in conformation with the result of Kiani et al.11, who reported that bacterial inoculation improved root length of sunflower up to 40% as compared to control under salt stressed conditions. Similarly12 recorded that inoculated treatment with Azospirillum brasilense significantly increased root length of maize as compared to un-inoculated control. The PGPR being free living organism facilitates rooting directly or indirectly. The result of this study showed more extended growth of roots that might be due to phytohormones produced by PGPR isolates. Indoleacetic acid and gibberellic acid in small quantities enhanced root growth. Vigorous root growth also stimulated nutrient uptake. The result of this study also confirmed the finding that significant effect of varieties and bio-inoculants and worked out with the conclusion that results were more marked in the root than in the shoot13.
During the present investigation, the number of tillers, total biomass and grain yield of wheat were increased by the application of PGPR alone and in combination (co-inoculation) of PGPR. Highest values were observed in mixture of WPR-32, WPR-42 and WPR-51 at 100 N.
Total biomass and grain yield was increased 83% and 54% as compared to control, respectively. The results were in line with the findings of Khalid et al.14, who reported that PGPR inoculation significantly enhanced number of tillers from 22-32% as compared to un inoculated. Total biomass and yield values were correlated. Treatments with higher total biomass values were also higher yielding treatment i.e., in mixture of three PGPR. These results were in concomitant with the findings of Wu et al.15 where 2-type of biofertilizer were used with control, chemical fertilizer and organic fertilizer. The application of three species of bacteria significantly increased the growth of maize and resulted in highest biomass that indicated half amount of bio fertilizer application had similar effects when compared with organic fertilizer treatments. The total biomass and yield was increased with the addition of nitrogen fertilizer as compared to control treatment16 also mentioned that PGPR enhanced plant growth, seed emergence and overall yield.
The results from this study revealed that shoot length was increased by PGPR at 0 N75 and 100 N. It was lowest at 0 N and almost doubled in 75 and 100% N. At harvesting stage the comparison of means showed non-significant differences among all the treatments except in mixture of WPR-32, WPR-42 and WPR-51. The treatments of 75 N and 100 N were at par with each other. Highest values were recorded in mixture and this is further reflected in high yield. These outcomes were in accordance with the findings of Ozturk et al.17, who suggested that combined inoculation of PGPR strains gave good response in equivalence to fertilizer treatment plants. The inoculation of PGPR strains increased wheat plant height due to inoculation with PGPR strains18. The PGPR influence crop growth and development by changing the physiological status19 and morphological characteristics of inoculated roots20. Similarly Tien et al.21, reported that increase in growth of wheat plants by PGPR was due to improve in N-use efficiency and production of phytohormones. Plant height despite of the fact that it is heredity controlled character least affected by other environmental factors but these factors in proper accordance may account for positive trend towards growth of crop especially plant height. Shoot dry weight was increased by PGPR inoculation. These results showed that the best way of increasing vigour of wheat crop or dry matter yield is through combined application of bio fertilizers as well as chemical fertilizers. These results were in line with the finding that interactive effect of organic and mineral fertilization of faba bean and wheat productivity achieved the increase in dry matters of wheat22,23.
During the present investigation, the number of tillers, total biomass and grain yield of wheat were increased by the application of PGPR alone and in combination (co-inoculation) of PGPR. Highest values were observed in mixture of WPR-32, WPR-42 and WPR-51 at 100 N. Total biomass and grain yield was increased 83% and 54% as compared to control respectively. The results were in line with the findings of Khalid et al.14, who reported that PGPR inoculation significantly enhanced number of tillers from 22-32% as compared to un inoculated control. Total biomass and yield values were correlated. Treatments with higher total biomass values were also higher yielding treatment i.e., in mixture of three PGPR. These results were in concomitant with the findings of Wu et al.15, where control, chemical fertilizer, organic fertilizer and 2-type of bio fertilizer were used. The application of three species of bacteria significantly increased the growth of maize and resulted in highest biomass. He also indicated that half the amount of bio fertilizer application had similar effects when compared with organic fertilizer treatments. The total biomass and yield was increased with the addition of nitrogen fertilizer as compared to control treatment.
It was reported that inoculation led to increase in number of grains/spike, 100 grain weight, grain and biological yield per plant and also concluded that bio-inoculation of PGPR strains led to increase in viable counts of PGPR in wheat rhizosphere. These strains complement each other and improved plant growth24.
Synergistic affects of multi-microbes on each other and on wheat was also reported by Khan and Zaidi25. The increased in number of tillers and yield in wheat crop due to inoculation of bio fertilizers also observed by Sarwar et al.26.
Plant growth promoting rhizo bacteria have ability to produced phytohormones could be attributed to their different genetic makeup, growth kinetics and enzymatic activities involved in auxin synthesis under given cultural conditions. These results were in conformity to the findings of some workers27.
Grain nitrogen was increased by PGPR application at three nitrogen levels, whereas results indicated in present investigation revealed that grain protein has not been much changed. These results revealed that integrated application of chemical fertilizers and bio fertilizers present best increase of nitrogen concentration required by plants for healthy and vegetative growth. These results were in line with28, who studied that inoculation with PGPR improved the maize growth and yield. The results also elucidated that nitrogen contents of can be maintained by applying nitrogen rich sources like chemical fertilizers and bio fertilizers.
The results revealed that PGPR and N fertilizer increase N, P, K contents of plants, whereas N, P, K of soil is decreased. It might be due to N and P uptake by plants is increased by the application of PGPR. The N, P and K levels of soil decreased with the application of PGPR and N fertilizer. This might be due to the increased utilization of soil nutrients by plants following the inoculation of PGPR. Application of PGPR enhanced uptake of nutrient elements like Ca, K, Fe, Cu, Mn and Zn by plants through stimulation of proton pump ATPase29,30 reported that combine the application of Bacillus and Microbacterium improves the uptake of the mineral elements by crop plants31 and 50% reduction in inorganic fertilizers because of microbial inoculation.
Beneficial effects of PGPR inoculants on plant growth and especially of root could be due to the result of multifarious mechanisms such as the production of siderophores, antibiotics, extra cellular metabolites (enzymes, vitamins) and induced systemic resistance, in addition to direct influence such as production of plant growth regulators, synthesis of extracellular enzymes to hydrolyze the fungal cell wall and competition for niches within the rhizosphere32.
This study implies that the inorganic fertilizers are not only costly but their use also causes environmental pollution, bio fertilizers can reduce the cost of inorganic fertilizers and also environment friendly. Results recommended that a combine application of PGPR WPR-32, WPR-42 and WPR-51 can reduces the use of inorganic fertilizer up to 25%.
CONCLUSION
It was concluded from this study that combined inoculation of PGPR WPR-32, WPR-42, and WPR-51 at 75 N gave the grain yield at par to 100 N. Hence it is recommended that the application of these microbes in the form of bio fertilizer can reduce the use of nitrogen fertilizer (by at least 25%) as well as save environmental pollution caused by fertilizer usage.
SIGNIFICANCE STATEMENT
Biofertilizers are very cost-effective and eco-friendly while, chemicals fertilizer are not only costly but also pollute our natural resources. The objective of this study was to search out a better combination of PGPR with chemical fertilizer that can reduce the inorganic fertilizer requirement for wheat crops. As a result of this study, it is concluded that nitrogen fertilizer application can be reduced by at least 25% without compromising the yield of wheat crops.
REFERENCES
- Vessey, J.K., 2003. Plant growth promoting rhizobacteria as biofertilizers. Plant Soil, 255: 571-586.
CrossRefDirect Link - Jacoby, R., M. Peukert, A. Succurro, A. Koprivova and S. Kopriva, 2017. The role of soil microorganisms in plant mineral nutrition-current knowledge and future directions. Front. Plant Sci., Vol. 8.
CrossRefDirect Link - Hussain, M.I., H.N. Asghar, M.J. Akhtar and M. Arshad, 2013. Impact of phosphate solubilizing bacteria on growth and yield of maize. Soil Environ., 32: 71-78.
Direct Link - Haas, D. and G. Defago, 2005. Biological control of soil-borne pathogens by fluorescent pseudomonads. Nat. Rev. Microbiol., 3: 307-319.
CrossRefPubMedDirect Link - Ashraf, M.A., M. Asif, A. Zaheer, A. Malik, Q. Ali and M. Rasool, 2013. Plant growth promoting rhizobacteria and sustainable agriculture: A review. Afr. J. Microbiol. Res., 7: 704-709.
CrossRefDirect Link - Steckel, J.E. and R.L. Flannery, 1971. Simultaneous Determinations of Phosphorus, Potassium, Calcium, and Magnesium in Wet Digestion Solutions of Plant Tissue by AutoAnalyzer. In: Instrumental Methods for Analysis of Soils and Plant Tissue, Walsh, L.M. (Ed.), Soil Science Society of America, Madison, WI, ISBN: 9780891187530, pp: 83-96.
CrossRefDirect Link - Murphy, J. and J.P. Riley, 1962. A modified single solution method for the determination of phosphate in natural waters. Anal. Chim. Acta, 27: 31-36.
CrossRefDirect Link - Kloepper, J.W., F.M. Scher, M. Laliberté and B. Tipping, 1986. Emergence-Promoting Rhizobacteria: Description and Implications for Agriculture. In: Iron, Siderophores, and Plant Diseases, Swinburne, T.R. (Ed.), Springer, Boston, MA, ISBN: 978-1-4615-9482-6, pp: 155-164.
CrossRefDirect Link - Glick, B.R., 1995. The enhancement of plant growth by free-living bacteria. Can. J. Microbiol., 41: 109-117.
CrossRefDirect Link - Kiani, M.Z., T. Sultan, A. Ali, I.A. Mahmood, T. Tabassam, M. Arshad Ullah and N. Abbas, 2016. Effect of PGPR strains on sunflower growth and nutrient contents under salinity stress. Pak. J. Agric. Res., 29: 141-148.
Direct Link - Fallik, E., Y. Okon, E. Epstein, A. Goldman and M. Fischer, 1989. Identification and quantification of IAA and IBA in Azospirillum brasilense-inoculated maize roots. Soil Biol. Biochem., 21: 147-153.
CrossRefDirect Link - Manske, G.G.B., J.I. Qritz-Monasterio, M. van Ginklel, R.M. Gozzalez, S. Rajaram, E. Molina and P.L.G. Vlek, 2000. Traits associated with improved P-uptake efficiency in CIMMYT's semi dwarf spring bread wheat grown on an acid Andisol in Mexico. Plant Soil, 221: 189-204.
CrossRefDirect Link - Khalid, A., M. Arshad and Z.A. Zahir, 2004. Screening plant growth-promoting rhizobacteria for improving growth and yield of wheat. J. Appl. Microbiol., 96: 473-480.
CrossRefDirect Link - Wu, S.C., Z.H. Cao, Z.G. Li, K.C. Cheung and M.H. Wong, 2005. Effects of biofertilizer containing N-fixer, P and K solubilizers and AM fungi on maize growth: A greenhouse trial. Geoderma, 125: 155-166.
CrossRefDirect Link - Ozturk, A., O. Caglar and F. Sahin, 2003. Yield response of wheat and barley to inoculation of plant growth promoting rhizobacteria at various levels of nitrogen fertilization. J. Plant Nutr. Soil Sci., 166: 262-266.
CrossRefDirect Link - Ashraf, M., S. Hasnain, O. Berge and T. Mahmood, 2004. Inoculating wheat seedlings with exopolysaccharide-producing bacteria restricts sodium uptake and stimulates plant growth under salt stress. Biol. Fertil. Soils, 40: 157-162.
CrossRefDirect Link - Glick, B.R. and Y. Bashan, 1997. Genetic manipulation of plant growth-promoting bacteria to enhance biocontrol of phytopathogens. Biotechnol. Adv., 15: 353-378.
CrossRefDirect Link - Grover, M., S. Bodhankar, A. Sharma, P. Sharma, J. Singh and L. Nain, 2021. PGPR mediated alterations in root traits: Way toward sustainable crop production. Front. Sustainable Food Syst., Vol. 4.
CrossRefDirect Link - Tien, T.M., M.H. Gaskins and D.H. Hubbell, 1979. Plant growth substances produced by Azospirillum brasilense and their effect on growth of pearl millet (Pennisetum americanum L.). Appl. Environ. Microbiol., 37: 1016-1024.
CrossRefDirect Link - Saleemi, M., M.Z. Kiani, T. Sultan, A. Khalid and S. Mahmood, 2017. Integrated effect of plant growth-promoting rhizobacteria and phosphate-solubilizing microorganisms on growth of wheat (Triticum aestivum L.) under rainfed condition. Agric. Food Secur., Vol. 6.
CrossRefDirect Link - Hilali, A., D. Prévost, W.J. Broughton and H. Antoun, 2011. Effects of inoculation with strains of Rhizobium leguminosarum biovar trifolii on the growth of wheat in two Moroccan soils. Can. J. Microbiol., 47: 590-593.
CrossRefDirect Link - Behl, R.K., H. Sharma, V. Kumar and N. Narula, 2003. Interactions amongst mycorrhiza, Azotobacter chroococcum and root characteristics of wheat varieties. J. Agron. Crop Sci., 189: 151-155.
CrossRefDirect Link - Khan, M.S. and A. Zaidi, 2007. Synergistic effects of the inoculation with plant growth-promoting rhizobacteria and an arbuscular mycorrhizal fungus on the performance of wheat. Turk. J. Agric. For., 31: 355-362.
Direct Link - Sarwar, N., Atique-ur-Rehman, O. Farooq, Allah Wasaya and M. Hussain et al., 2021. Integrated nitrogen management improves productivity and economic returns of wheat-maize cropping system. J. King Saud Univ. Sci., Vol. 33.
CrossRefDirect Link - Khalid, A., M. Arshad and Z.A. Zahir, 2001. Factors affecting auxin biosynthesis by wheat and rice rhizobacteria. Pak. J. Soil Sci., 20: 11-18.
Direct Link - Arshad, M. and W.T. Frankenberger Jr., 1997. Plant growth-regulating substances in the rhizosphere: Microbial production and functions. Adv. Agron., 62: 45-151.
CrossRefDirect Link - Mantelin, S. and B. Touraine, 2004. Plant growth-promoting bacteria and nitrate availability: Impacts on root development and nitrate uptake. J. Exp. Bot., 55: 27-34.
CrossRefDirect Link - Karlidag, H., A. Esitken, M. Turan and F. Sahin, 2007. Effects of root inoculation of plant growth promoting rhizobacteria (PGPR) on yield, growth and nutrient element contents of leaves of apple. Sci. Hortic., 114: 16-20.
CrossRefDirect Link - Zahir, Z.A., M. Arshad and W.T. Frankenberger Jr., 2004. Plant growth promoting rhizobacteria: Applications and perspectives in agriculture. Adv. Agron., 81: 97-168.
CrossRefDirect Link - Yanni, Y.G., R.Y. Rizk, V. Corich, A. Squartini and K. Ninke et al., 1997. Natural endophytic association between Rhizobium leguminosarum bv. trifolii and rice roots and assessment of its potential to promote rice growth. In: Opportunities for Biological Nitrogen Fixation in Rice and Other Non-Legumes, Ladha, J.K., F.J. Bruijn and K.A. Malik (Eds.), Springer, Dordrecht, ISBN: 978-0-7923-4748-4, pp: 99-114.
CrossRefDirect Link